Validating MsrB1 Enzyme Inhibitors: A Comprehensive Guide to the NADPH Consumption Assay
This article provides researchers and drug development professionals with a comprehensive guide to using the NADPH consumption assay for validating methionine sulfoxide reductase B1 (MsrB1) inhibitors.
Validating MsrB1 Enzyme Inhibitors: A Comprehensive Guide to the NADPH Consumption Assay
Abstract
This article provides researchers and drug development professionals with a comprehensive guide to using the NADPH consumption assay for validating methionine sulfoxide reductase B1 (MsrB1) inhibitors. We explore the foundational role of MsrB1 in oxidative stress and disease, detail the step-by-step methodology of the assay, address common troubleshooting and optimization challenges, and discuss validation strategies and comparative analysis with other techniques. The content synthesizes current knowledge to offer a practical framework for robust inhibitor screening in biomedical research.
MsrB1, Oxidative Stress, and the Rationale for Targeted Inhibition
Introduction & Biological Context Methionine Sulfoxide Reductase B1 (MsrB1) is a key selenoprotein responsible for the stereospecific reduction of methionine-R-sulfoxide residues in proteins back to methionine. This repair function is critical in reversing oxidative damage, thereby regulating protein function, mitigating oxidative stress, and influencing cellular signaling pathways. Within the context of NADPH consumption assay-based inhibitor validation research, MsrB1 is a significant therapeutic target. Its inhibition is explored for conditions where modulating redox signaling is beneficial, such as in certain cancers or age-related diseases. Inhibitor validation relies on assays measuring the enzymatic consumption of NADPH, a crucial cofactor in the MsrB1 catalytic cycle.
Key Functions and Quantifiable Impact The biological significance of MsrB1 is demonstrated through its quantifiable effects on cellular and organismal physiology. The table below summarizes key experimental findings.
Table 1: Quantified Biological Impacts of MsrB1 Modulation
| Parameter Measured | Experimental Condition | Quantitative Result / Change | Biological Implication |
|---|---|---|---|
| Intracellular ROS | MsrB1 Knockdown (e.g., siRNA) | Increase of 40-60% in H₂O₂-induced cells | Highlights role in antioxidant defense. |
| Protein Carbonyls (Oxidation Marker) | MsrB1 Overexpression | Decrease of ~35% vs. control under oxidative stress | Confirms protein repair/protective function. |
| NADPH Consumption Rate | In vitro Recombinant MsrB1 Activity | 0.8 - 1.2 µmol NADPH/min/mg enzyme | Baseline for inhibitor efficacy screening. |
| Cell Viability (e.g., Cancer Line) | MsrB1 Inhibition + Chemotherapy | Synergistic decrease; IC₅₀ reduced by 50-70% | Supports combination therapy potential. |
| Target Engagement (IC₅₀) | Candidate Inhibitor in NADPH Assay | Ranges from nM to µM concentrations | Primary metric for inhibitor potency ranking. |
The Scientist's Toolkit: Research Reagent Solutions Table 2: Essential Reagents for MsrB1 & NADPH Consumption Assay Research
| Reagent / Material | Function / Purpose |
|---|---|
| Recombinant Human MsrB1 Protein | Purified enzyme source for in vitro kinetic and inhibitor screening assays. |
| NADPH (Tetrasodium Salt) | Essential reducing cofactor; its oxidation is monitored at 340 nm to quantify enzyme activity. |
| Dithiothreitol (DTT) or TCEP | Thiol-based reducing agents required to regenerate the active site selenol/thiol of MsrB1. |
| Methionine-R-Sulfoxide (Met-R-SO) | Stereospecific substrate for MsrB1. Critical for activity assays. |
| Candidate Small-Molecule Inhibitors | Compounds for validation; typically from virtual screening or structural analogs. |
| 96/384-Well UV-Transparent Microplates | Plate format for high-throughput spectrophotometric NADPH consumption assays. |
| Specific MsrB1 Antibodies (e.g., ELISA/SDS-PAGE) | For quantifying protein expression, pull-down assays, or cellular target engagement studies. |
| siRNA/shRNA for MSRB1 Gene | For genetic knockdown to study loss-of-function phenotypes in cell models. |
Experimental Protocol 1: Core NADPH Consumption Assay for MsrB1 Inhibitor Screening This protocol is the cornerstone for validating and characterizing MsrB1 inhibitors.
Objective: To measure the initial rate of NADPH oxidation catalyzed by MsrB1 in the presence of a test compound to determine inhibitory potency (IC₅₀).
Materials:
- Assay Buffer: 50 mM HEPES, pH 7.5, 150 mM NaCl.
- Recombinant MsrB1 (final conc. 5-20 nM).
- NADPH (final conc. 200 µM).
- DTT (final conc. 5 mM).
- Substrate: Met-R-SO (final conc. 1-5 mM).
- Test compounds/inhibitors dissolved in DMSO (<1% final DMSO).
- UV-transparent 96-well plate.
- Plate reader capable of kinetic measurements at 340 nm.
Procedure:
- Master Mix Preparation: In a tube, combine Assay Buffer, NADPH, and DTT. Keep on ice.
- Enzyme-Inhibitor Pre-incubation: In the reaction wells, add MsrB1 to the master mix. Immediately add test compound at varying concentrations (e.g., 0.1 nM - 100 µM). Include negative control (DMSO vehicle) and blank (no enzyme).
- Initiation: Pre-incubate plate at 25°C for 10-15 min in the plate reader. Initiate the reaction by automated injection of Met-R-SO substrate.
- Kinetic Measurement: Immediately monitor the decrease in absorbance at 340 nm (A₃₄₀) for 10-15 minutes at 25°C.
- Data Analysis: Calculate the slope (ΔA₃₄₀/min) for the initial linear phase for each well. Subtract the blank rate. Express activity as a percentage of the vehicle control (100% activity). Fit inhibitor dose-response data to a sigmoidal curve to determine IC₅₀ values.
Experimental Protocol 2: Cellular Target Engagement via Immunoprecipitation & MSR Activity Objective: To confirm that a candidate inhibitor engages with and inhibits cellular MsrB1.
Materials:
- Cultured cells (relevant to disease model).
- Cell lysis buffer (with protease inhibitors, without strong reductants).
- Candidate inhibitor and vehicle control.
- Anti-MsrB1 antibody for immunoprecipitation (IP).
- Protein A/G beads.
- Standard NADPH consumption assay reagents (as in Protocol 1).
Procedure:
- Cell Treatment: Treat cells with inhibitor or vehicle for a predetermined time (e.g., 4-24 h).
- Cell Lysis & IP: Lyse cells, clarify lysate. Incubate equal protein amounts with anti-MsrB1 antibody, then with Protein A/G beads. Wash beads thoroughly.
- On-Bead Activity Assay: Resuspend the IP beads directly in the NADPH assay master mix (containing NADPH, DTT, and buffer) in a microplate well.
- Activity Measurement: Initiate reaction by adding Met-R-SO. Monitor A₃₄₀ kinetically. Compare the activity of MsrB1 immunoprecipitated from inhibitor-treated cells versus vehicle-treated cells.
- Analysis: A significant reduction in activity from the inhibitor-treated sample confirms cellular target engagement, independent of changes in MsrB1 protein levels (which can be verified by Western blot from the same IP eluate).
Visualization of Concepts and Workflows
Diagram 1: MsrB1 Function & Inhibition Logic
Diagram 2: Inhibitor Screening Assay Steps
Application Notes
MsrB1 (Methionine Sulfoxide Reductase B1) is a key selenium-dependent enzyme responsible for the reduction of methionine-R-sulfoxide back to methionine, a critical repair mechanism for proteins damaged by reactive oxygen species (ROS). Its activity is intrinsically linked to cellular redox homeostasis. Within the context of validating MsrB1 inhibitors via NADPH consumption assays, understanding its multifaceted role in disease pathologies is crucial for target rationale and inhibitor characterization.
- Cancer Context: MsrB1 exhibits a dual role in cancer. In many solid tumors (e.g., liver, gastric), MsrB1 is overexpressed, protecting cancer cells from oxidative stress, promoting proliferation, and contributing to chemoresistance. Inhibiting MsrB1 in these contexts could sensitize tumors to oxidative damage-induced cell death. Conversely, in some hematological cancers, it may act as a tumor suppressor. NADPH consumption assays on cell lysates from treated tumor models can quantify the effective enzymatic inhibition of candidate compounds, correlating it with increased oxidative stress markers and reduced cell viability.
- Neurodegeneration Context: In diseases like Alzheimer's and Parkinson's, oxidative damage to neuronal proteins is a hallmark. MsrB1 is vital for maintaining the function of proteins like tau and α-synuclein by repairing methionine oxidation. Its activity often declines with age. Inhibiting MsrB1 is not the therapeutic goal here; rather, research focuses on its upregulation or protection. However, NADPH consumption assays are essential tools to study the impact of disease-related post-translational modifications or mutations on MsrB1's enzymatic efficiency in brain tissue samples.
- Aging Context: The age-associated decline in MsrB1 function contributes to the accumulation of oxidized proteins, driving cellular senescence and tissue dysfunction. Research into MsrB1 activators or mimetics is a key anti-aging strategy. In inhibitor validation research for other diseases, the NADPH consumption assay serves as a critical control to ensure candidate compounds do not inadvertently impair MsrB1 in healthy tissues, which could accelerate aging-related phenotypes.
Table 1: Quantitative Summary of MsrB1 Dysregulation in Disease Models
| Disease Category | Model System | Observed Change in MsrB1 (vs. Control) | Key Functional Consequence | Assay for Validation |
|---|---|---|---|---|
| Hepatocellular Carcinoma | Human tumor tissue | Up to 3.5-fold increase (mRNA & protein) | Enhanced tumor growth, reduced ROS | NADPH consumption in lysates; cell viability post-inhibition |
| Alzheimer's Disease | AD mouse model (e.g., 3xTg) | ~40-60% decrease in hippocampal protein | Accumulation of oxidized tau, synaptic loss | NADPH consumption assay on brain homogenates |
| Aging | Aged mouse liver (24mo vs 3mo) | ~50% decrease in enzyme activity | Increased protein carbonyls, metabolic decline | Direct NADPH-coupled enzyme activity assay |
Experimental Protocols
Protocol 1: Recombinant MsrB1 Purification for Biochemical Assays
Purpose: To obtain pure, active MsrB1 enzyme for high-throughput inhibitor screening and kinetic studies.
- Cloning & Expression: Clone human MSRB1 gene into a pET vector with an N-terminal His-tag. Transform into E. coli BL21(DE3) cells.
- Induction: Grow culture in LB + antibiotic to OD600 ~0.6. Induce with 0.5 mM IPTG at 18°C for 16 hours.
- Lysis: Harvest cells by centrifugation. Resuspend pellet in Lysis Buffer (50 mM Tris-HCl pH 7.5, 300 mM NaCl, 10 mM imidazole, protease inhibitors). Lyse by sonication on ice.
- Purification: Clarify lysate by centrifugation. Load supernatant onto a Ni-NTA affinity column pre-equilibrated with Lysis Buffer.
- Wash & Elution: Wash with 20 column volumes of Wash Buffer (50 mM Tris-HCl pH 7.5, 300 mM NaCl, 25 mM imidazole). Elute with Elution Buffer (Wash Buffer with 250 mM imidazole).
- Dialysis & Storage: Dialyze eluted protein into Storage Buffer (50 mM HEPES pH 7.0, 100 mM NaCl, 10% glycerol). Determine concentration, aliquot, and store at -80°C. Assess purity by SDS-PAGE.
Protocol 2: NADPH-Coupled MsrB1 Activity & Inhibition Assay
Purpose: To measure MsrB1 enzymatic activity and determine the IC₅₀ of inhibitors by monitoring NADPH oxidation.
- Reaction Setup: In a clear 96-well plate, mix the following for a 100 µL final volume per well:
- Assay Buffer: 50 mM HEPES pH 7.5, 50 mM NaCl, 10 mM DTT.
- Coupling System: 0.25 mM NADPH, 0.5 U/mL Thioredoxin Reductase (from rat liver), 5 µM E. coli Thioredoxin.
- Substrate: 2 mM Dabsyl-Met-R-Sulfoxide (synthetic substrate).
- Inhibitor: Serial dilutions of the test compound in DMSO (include DMSO-only controls).
- Enzyme: Initiate reaction by adding purified recombinant MsrB1 (final 10-50 nM).
- Kinetic Measurement: Immediately monitor the decrease in absorbance at 340 nm (A₃₄₀) for NADPH consumption using a plate reader at 30°C for 10-15 minutes.
- Data Analysis: Calculate the initial reaction rate (V₀) from the linear slope of A₃₄₀ vs. time. Normalize activity of inhibitor wells to the vehicle control (100% activity). Plot % activity vs. inhibitor concentration and fit with a sigmoidal dose-response curve to calculate IC₅₀.
Protocol 3: Cellular MsrB1 Inhibition Validation Using Lysate NADPH Consumption
Purpose: To confirm target engagement of MsrB1 inhibitors in a cellular context.
- Cell Treatment: Treat relevant cell lines (e.g., cancer lines) with the candidate inhibitor or vehicle for 6-24 hours.
- Lysate Preparation: Wash cells with PBS, then lyse in Assay Buffer (see Protocol 2) without DTT, using freeze-thaw cycles or gentle detergent.
- Clarification: Centrifuge lysates at 15,000 x g for 10 min at 4°C to remove debris. Keep supernatants on ice.
- Coupled Assay: Perform the NADPH-coupled assay as in Protocol 2, substituting recombinant enzyme with a standardized amount of total cellular protein (e.g., 20 µg) from the lysates.
- Interpretation: A concentration-dependent decrease in NADPH consumption rate in inhibitor-treated lysates confirms cellular penetration and target engagement. Compare to recombinant enzyme IC₅₀.
Visualizations
Title: MsrB1 Repair Cycle & Inhibitor Target
Title: NADPH-Coupled MsrB1 Inhibitor Assay Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for MsrB1/NADPH Consumption Research
| Item | Function & Rationale |
|---|---|
| Recombinant Human MsrB1 (His-tagged) | Pure, active enzyme for primary biochemical screening and kinetic analysis. His-tag allows easy purification and immobilization. |
| Dabsyl-Methionine-R-Sulfoxide | Synthetic, chromogenic substrate. MsrB1 specifically reduces the R-sulfoxide, enabling direct coupling to the NADPH/Thioredoxin system. |
| NADPH (Tetrasodium Salt) | Essential reducing cofactor. Its oxidation is measured at 340 nm to quantify the coupled enzymatic activity. |
| Rat Liver Thioredoxin Reductase | The coupling enzyme that reduces oxidized thioredoxin using NADPH, driving the cyclic reaction and amplifying the signal. |
| E. coli Thioredoxin | Electron donor for MsrB1. It is oxidized during the reaction and subsequently recycled by Thioredoxin Reductase. |
| DTT (Dithiothreitol) | Maintains reducing environment in enzyme storage and assay buffers to keep MsrB1 active. |
| HEPES Buffer (pH 7.5) | Provides optimal pH stability for the coupled enzyme system during kinetic measurements. |
| 96-Well Clear UV-Transparent Plates | Compatible with spectrophotometric detection at 340 nm for high-throughput format. |
| Microplate Reader with Kinetic Capability | For automated, simultaneous monitoring of NADPH consumption (A₃₄₀) over time in multiple samples. |
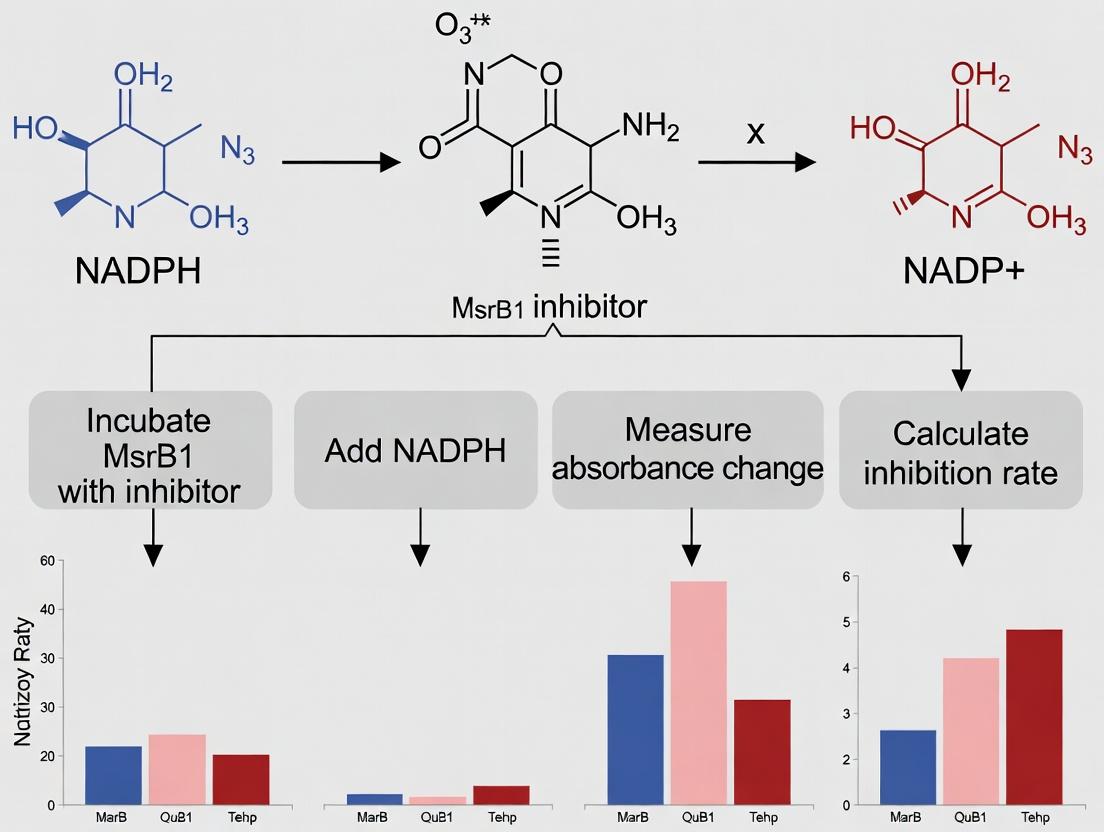
Introduction Within the context of validating inhibitors for the methionine sulfoxide reductase B1 (MsrB1) enzyme, the NADPH consumption assay is a cornerstone functional readout. MsrB1 catalyzes the reduction of methionine-R-sulfoxide residues in proteins, a critical antioxidant repair pathway. This enzymatic activity is coupled to the thioredoxin (Trx) system, ultimately leading to the oxidation of NADPH to NADP⁺. Monitoring the depletion of NADPH spectrophotometrically provides a direct, quantitative measure of MsrB1 enzymatic velocity. Inhibitor validation hinges on detecting a concentration-dependent decrease in this consumption rate. This application note details the biochemical rationale, protocols, and key reagents for implementing this assay in drug discovery research.
Biochemical Pathway and Assay Principle MsrB1 activity is regenerated through a series of redox transfers originating from NADPH. The diagram below illustrates this coupled electron flow.
Diagram Title: NADPH-Coupled Electron Transfer Pathway for MsrB1 Activity
Quantitative Data Summary Key kinetic parameters for MsrB1 and related system components inform assay design. The following table summarizes typical values.
Table 1: Representative Kinetic Parameters for the MsrB1/Thioredoxin System
| Component | Parameter | Typical Value | Notes & Experimental Conditions |
|---|---|---|---|
| MsrB1 (Human) | Km for DTT* (mM) | 8.5 - 12.0 | *Direct reductant in simplified assays. |
| MsrB1 (Human) | Km for Trx (µM) | 25 - 40 | Physiological reductant. |
| Thioredoxin (Trx1) | Km for TrxR (µM) | 3 - 6 | Varies by isoform and species. |
| NADPH | Extinction Coefficient (ε340) | 6220 M⁻¹cm⁻¹ | Fundamental for consumption calculation. |
| MsrB1 Activity | Specific Activity | 15 - 25 nmol/min/mg | Using dabsyl-MetRO substrate. |
| Assay Signal | ΔA340/min (Signal Window) | 0.05 - 0.15 | For robust quantitation in 100 µL assay. |
*DTT: Dithiothreitol, often used as a non-physiological reductant in initial characterization.
Research Reagent Solutions Toolkit Table 2: Essential Materials for NADPH-Consumption MsrB1 Inhibitor Assay
| Reagent / Solution | Function & Rationale |
|---|---|
| Recombinant Human MsrB1 | Purified catalytic enzyme; source of activity. |
| Recombinant Thioredoxin (Trx1) & Thioredoxin Reductase (TrxR1) | Complete physiological redox relay system. |
| β-NADPH, Tetrasodium Salt | Essential redox cofactor and assay substrate. Primary signal source (A340). |
| Methionine-R-Sulfoxide (MetRO) Substrate | Natural substrate (e.g., dabsyl-MetRO for sensitivity, or protein-bound). |
| Candidate Inhibitor Compounds | Dissolved in DMSO; final DMSO concentration must be standardized (e.g., ≤1%). |
| Assay Buffer (Tris or Phosphate, pH 7.4-7.8) | Provides optimal physiological pH for enzyme activity. |
| EDTA (0.5-1 mM) | Chelating agent to inhibit metal-dependent oxidation. |
| BSA (0.1 mg/mL) | Stabilizes low-concentration enzymes, reduces non-specific binding. |
| Microplate Reader (UV-Vis, 340 nm) | Instrument for kinetic absorbance measurement. |
| Clear 96- or 384-Well Plates | Compatible format for high-throughput screening. |
Detailed Experimental Protocol
Protocol 1: Direct, Coupled NADPH Consumption Assay for MsrB1 Inhibitor Screening
I. Primary Workflow The high-level experimental process is visualized below.
Diagram Title: MsrB1 NADPH Consumption Assay Workflow
II. Materials & Reagent Preparation
- MsrB1 Enzyme Solution: Dilute recombinant MsrB1 in assay buffer with 0.1 mg/mL BSA to 2x final desired concentration (e.g., 20-50 nM final).
- Redox System Solution: Combine TrxR (e.g., 50 nM final), Trx (e.g., 5 µM final) in assay buffer. Keep on ice.
- NADPH Solution: Prepare fresh in assay buffer (e.g., 150 µM final). Concentration should yield an initial A340 ~0.8-1.0 in the assay volume.
- Substrate Solution: Dissolve MetRO (e.g., dabsyl-MetRO) in assay buffer (e.g., 200-500 µM final).
- Inhibitor/Control Solutions: Prepare compound serial dilutions in 100% DMSO. Include a vehicle control (DMSO only).
III. Step-by-Step Procedure
- Assay Setup: In a clear, flat-bottom microplate, add 25 µL of substrate solution per well.
- Inhibitor Addition: Add 2.5 µL of inhibitor or DMSO control to appropriate wells. For no-enzyme background controls, add buffer instead.
- Enzyme/Redox System Addition: Add 12.5 µL of the Redox System Solution, followed by 10 µL of the MsrB1 Enzyme Solution to all sample wells. To background control wells, add equivalent volumes of buffer without enzyme.
- Volume Equilibration: Add assay buffer to bring the total volume in each well to 95 µL. Mix gently by pipetting or plate shaking.
- Pre-incubation: Incubate plate at 25°C or 37°C for 10-15 minutes to allow temperature equilibration and potential inhibitor binding.
- Reaction Initiation: Using a multichannel pipette, rapidly add 5 µL of the NADPH Solution to each well to start the reaction (final volume = 100 µL). Final DMSO concentration should be ≤1%.
- Kinetic Measurement: Immediately place the plate in a pre-warmed microplate reader. Monitor the decrease in absorbance at 340 nm (A340) every 30-60 seconds for 30-60 minutes.
IV. Data Analysis
- Raw Data Processing: For each well, plot A340 vs. time. Perform linear regression on the linear portion of the curve (typically first 10-20 minutes).
- Rate Calculation: The slope (ΔA340/min) is the reaction velocity (v). Convert to NADPH consumption rate using the Beer-Lambert law: Rate (nM/min) = (Slope / 6220 M⁻¹cm⁻¹) * (10⁹ nM/M) / Pathlength (cm). Pathlength is plate-specific.
- Background Subtraction: Subtract the rate from the no-enzyme control wells from all sample rates.
- Inhibition Calculation: For each inhibitor concentration, calculate percent inhibition: % Inhibition = [1 - (vinhibited / vcontrol)] * 100, where v_control is the rate with DMSO vehicle only.
- IC₅₀ Determination: Fit % Inhibition vs. log10[Inhibitor] data to a four-parameter logistic (4PL) curve to determine the half-maximal inhibitory concentration.
Methionine sulfoxide reductase B1 (MsrB1) is a key selenoprotein responsible for the stereospecific reduction of methionine-R-sulfoxide back to methionine, a critical repair mechanism for oxidative damage to proteins. This function positions MsrB1 as a central regulator of cellular redox homeostasis, protein function, and signal transduction. Inhibition of MsrB1 is being explored as a therapeutic strategy in diseases characterized by aberrant redox signaling and cellular proliferation, most notably in certain cancers. This document provides application notes and protocols for validating MsrB1 inhibitors, framed within a thesis focused on NADPH consumption assays for inhibitor screening and characterization.
Thesis Context: NADPH Consumption Assay in MsrB1 Inhibitor Validation
The core thesis posits that direct, real-time measurement of NADPH oxidation provides a robust, quantitative primary screen for MsrB1 enzymatic activity and its inhibition. As MsrB1 requires thioredoxin (Trx) and thioredoxin reductase (TrxR) for catalytic recycling, its activity is coupled to NADPH consumption. A decrease in the rate of NADPH oxidation in the presence of a test compound indicates potential inhibitor activity. This assay forms the foundational validation step before progressing to cell-based and mechanistic studies.
Application Notes & Protocols
Protocol 1: Recombinant Human MsrB1 Enzyme Purification
Objective: To obtain purified, active MsrB1 for in vitro assays. Materials:
- E. coli expression system (e.g., BL21(DE3)) harboring plasmid for His-tagged human MsrB1.
- LB media supplemented with antibiotics, selenocysteine precursors (e.g., sodium selenite).
- Lysis Buffer: 50 mM Tris-HCl (pH 8.0), 300 mM NaCl, 10 mM imidazole, 0.1% Triton X-100, protease inhibitors.
- Elution Buffer: 50 mM Tris-HCl (pH 8.0), 300 mM NaCl, 250 mM imidazole.
- Storage Buffer: 50 mM Tris-HCl (pH 7.5), 150 mM NaCl, 10% glycerol.
- Ni-NTA affinity chromatography column. Procedure:
- Induce expression with IPTG at OD600 ~0.6-0.8.
- Harvest cells by centrifugation and lyse via sonication in Lysis Buffer on ice.
- Clarify lysate by centrifugation at 15,000 x g for 30 min.
- Load supernatant onto a pre-equilibrated Ni-NTA column.
- Wash with 10-20 column volumes of Lysis Buffer.
- Elute bound protein with Elution Buffer.
- Desalt into Storage Buffer using a PD-10 column or dialysis.
- Determine concentration (A280), aliquot, and store at -80°C.
Protocol 2: Direct, Coupled NADPH Consumption Assay
Objective: To measure MsrB1 enzymatic activity and inhibitor IC50 by monitoring NADPH oxidation. Principle: The assay couples MsrB1 activity to the oxidation of NADPH via the endogenous thioredoxin (Trx) and thioredoxin reductase (TrxR) system. Inhibition of MsrB1 reduces the consumption rate of NADPH, measured by a decrease in absorbance at 340 nm. Reaction Setup (96-well plate format, final volume 100 µL):
- Assay Buffer: 50 mM HEPES (pH 7.5), 150 mM NaCl, 1 mM EDTA.
- Coupling System: 100 µM NADPH, 5 µM human Trx1, 100 nM human TrxR1.
- Substrate: 1-5 mM Dabsyl-Met-R-O (synthetic substrate) or 10 mM free Met-R-O.
- Enzyme: 50-100 nM purified recombinant human MsrB1.
- Inhibitor: Test compound (varying concentrations, DMSO concentration normalized ≤1%). Procedure:
- To each well, add Assay Buffer, NADPH, Trx, TrxR, and inhibitor or vehicle control. Pre-incubate for 5-10 min at 25°C.
- Initiate the reaction by adding MsrB1 enzyme.
- Immediately monitor the decrease in absorbance at 340 nm (A340) for 10-15 minutes using a plate reader.
- Calculate the initial velocity (Vo) from the linear portion of the curve (ΔA340/min).
- Plot Vo (or % Activity) against inhibitor concentration to determine IC50.
Table 1: Representative NADPH Consumption Assay Data for Candidate Inhibitors
| Compound ID | Chemical Class | IC50 (µM) | Hill Slope | % Activity at 100 µM | Assay Z'-factor |
|---|---|---|---|---|---|
| M1B-107 | Selenocysteine mimic | 0.15 ± 0.03 | 1.1 | 2.5 | 0.78 |
| ATO-216 | Reversible covalent | 2.4 ± 0.5 | 1.4 | 15.3 | 0.72 |
| Control | Known inhibitor | 1.8 ± 0.2 | 1.0 | 10.1 | - |
| DMSO | Vehicle | N/A | N/A | 100 | - |
Table 2: Key Research Reagent Solutions
| Reagent | Function/Explanation | Source/Example |
|---|---|---|
| Recombinant hMsrB1 | Catalytic enzyme; high purity is essential for accurate kinetic measurements. | In-house purification from E. coli or commercial (e.g., R&D Systems). |
| Trx/TrxR/NADPH System | Regenerating electron donor system; couples MsrB1 activity to measurable NADPH oxidation. | Commercial kits available (Cayman Chemical) or individual components. |
| Dabsyl-Met-R-O | Synthetic, chromogenic substrate; allows direct activity measurement without coupling system. | Custom synthesis required. |
| Selective MsrB Inhibitor (e.g., M1B-107) | Positive control for inhibition; validates assay sensitivity. | Literature compounds or in-house discovery. |
| LC-MS/MS System | For orthogonal validation of methionine sulfoxide reduction in complex protein substrates. | e.g., Agilent 6495C QQQ. |
| MDA-MB-231 Cell Lysate | Relevant biological matrix for testing inhibitor activity in a complex, disease-relevant background. | ATCC; derived from triple-negative breast cancer line. |
Protocol 3: Orthogonal Validation in Cellular Context
Objective: To confirm inhibitor activity in a complex biological system. Workflow:
- Cell Treatment: Treat relevant cancer cell lines (e.g., MDA-MB-231) with inhibitor for 24h.
- Lysate Preparation: Lyse cells in RIPA buffer + protease/phosphatase inhibitors.
- Exogenous Substrate Assay: Spike treated cell lysates with a known amount of recombinant MsrB1 and its substrate. Measure residual activity using Protocol 2.
- Western Blot Analysis: Probe for accumulation of protein-bound Met-R-O (using specific antibodies) and downstream targets (e.g., altered NF-κB, p53 signaling).
Visualizations
Diagram 1 Title: MsrB1 Inhibitor Validation Workflow
Diagram 2 Title: MsrB1 Catalytic Cycle & Assay Principle
Step-by-Step Protocol: Executing the NADPH Consumption Assay for MsrB1
This document details the application notes and protocols for a Methionine Sulfoxide Reductase B1 (MsrB1) enzyme activity assay, framed within a broader thesis on NADPH consumption assay for MsrB1 inhibitor validation. MsrB1 is a key enzyme responsible for the reduction of methionine-R-sulfoxide residues in proteins, playing a critical role in cellular antioxidant defense and redox regulation. Its inhibition is a promising therapeutic strategy for conditions involving oxidative stress, such as aging-related diseases and certain cancers. The assay quantifies MsrB1 activity by coupling the enzymatic reduction of its substrate to the oxidation of NADPH, which is monitored spectrophotometrically. This method is fundamental for screening and validating small-molecule inhibitors in drug development pipelines.
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in MsrB1 Assay |
|---|---|
| Recombinant MsrB1 Protein | The enzyme of interest. Catalyzes the reduction of methionine-R-sulfoxide, initiating the coupled reaction that consumes NADPH. |
| NADPH (Tetrasodium Salt) | The essential cofactor and signal molecule. Its oxidation to NADP+ is stoichiometrically linked to MsrB1 activity, and its decreasing concentration is measured at 340 nm. |
| DTNB (5,5'-Dithio-bis-(2-nitrobenzoic acid), Ellman's Reagent) | Used in a secondary assay to measure the concentration of free thiols (e.g., DTT) generated during the enzyme's catalytic cycle. |
| Dithiothreitol (DTT) | The physiological reductant that regenerates the active site of MsrB1, producing a measurable thiol byproduct. |
| Methionine-R-Sulfoxide (Met-R-SO) | The specific substrate for the MsrB1 enzyme. |
| Plate Reader (UV-Vis capable) | Instrument for high-throughput measurement of absorbance changes at 340 nm (NADPH) or 412 nm (DTNB-TNB) in multi-well plates. |
| Assay Buffer (e.g., Tris-HCl, pH 7.5) | Provides optimal ionic strength and pH for MsrB1 enzymatic activity. |
Protocols
Protocol 1: Primary NADPH Consumption Assay for MsrB1 Activity and Inhibition
Objective: To directly measure MsrB1 enzymatic activity by monitoring the decrease in NADPH absorbance at 340 nm, and to validate inhibitor efficacy.
Materials:
- Recombinant Human MsrB1 (≥95% purity)
- NADPH, tetrasodium salt
- Dithiothreitol (DTT)
- L-Methionine-R-Sulfoxide (Met-R-SO)
- Assay Buffer: 50 mM Tris-HCl, 150 mM NaCl, pH 7.5
- Putative small-molecule inhibitors (e.g., in DMSO)
- 96-well or 384-well clear-bottom microplates
- UV-Visible spectrophotometric plate reader
Procedure:
- Solution Preparation: Prepare all reagents in assay buffer on ice. A master mix containing MsrB1 (final reaction concentration: 50-100 nM) and NADPH (final: 100-200 µM) is recommended.
- Inhibitor Pre-incubation: For inhibitor testing, pre-incubate MsrB1 with the putative inhibitor (varying concentrations) in the plate for 15 minutes at 25°C. Include controls (vehicle-only, e.g., DMSO).
- Reaction Initiation: Start the reaction by adding a pre-mixed solution of DTT (final: 1-5 mM) and Met-R-SO (final: 0.5-2 mM). Final reaction volume is typically 100 µL.
- Kinetic Measurement: Immediately transfer the plate to a pre-warmed (37°C) plate reader. Measure the absorbance at 340 nm every 20-60 seconds for 30-60 minutes.
- Data Analysis: Calculate the initial velocity (V0) from the linear portion of the absorbance decrease. Plot V0 vs. inhibitor concentration to determine IC50 values.
Protocol 2: Coupled DTNB Assay for Thiol Quantification
Objective: To measure MsrB1 activity indirectly by quantifying the amount of DTT consumed (or TNB produced) using DTNB, serving as an orthogonal validation method.
Materials:
- All materials from Protocol 1.
- DTNB (Ellman’s Reagent) prepared fresh in assay buffer.
Procedure:
- Enzymatic Reaction: Perform the MsrB1 reaction as in Protocol 1 (Steps 1-3) in a tube, but without NADPH in the mix. Terminate the reaction after 10-30 minutes by heat inactivation (70°C for 5 min) or acidification.
- DTNB Development: Centrifuge the terminated reaction to pellet precipitated protein. Transfer an aliquot of the supernatant to a plate well.
- Color Development: Add DTNB to a final concentration of 0.2-1 mM. Incubate for 5-15 minutes at room temperature, protected from light.
- Measurement: Read the absorbance at 412 nm in a plate reader. The yellow-colored 2-nitro-5-thiobenzoate (TNB) product is proportional to the free thiol concentration (from unreacted DTT). Lower TNB signal indicates higher MsrB1 activity (more DTT consumed).
Data Presentation
Table 1: Representative Data from MsrB1 Inhibitor Screen Using NADPH Consumption Assay
| Inhibitor Compound | Concentration (µM) | ΔA340/min (V0) | % Activity Remaining | Calculated IC50 (µM) |
|---|---|---|---|---|
| Vehicle (DMSO) Control | 0 | -0.025 ± 0.002 | 100% | N/A |
| Compound A | 1 | -0.022 ± 0.001 | 88% | 12.5 ± 1.8 |
| Compound A | 10 | -0.012 ± 0.001 | 48% | 12.5 ± 1.8 |
| Compound A | 100 | -0.002 ± 0.0005 | 8% | 12.5 ± 1.8 |
| Compound B (Reference) | 10 | -0.001 ± 0.0003 | 4% | 0.85 ± 0.10 |
Table 2: Comparison of Primary and Orthogonal Assay Results
| Assay Type | Signal Measured | Wavelength | Key Advantage | Key Limitation |
|---|---|---|---|---|
| NADPH Consumption | Decrease in A340 (NADPH → NADP+) | 340 nm | Direct, continuous, kinetic measurement of the physiological redox couple. | Potential interference from compounds that absorb at 340 nm. |
| DTNB (Ellman's) | Increase in A412 (TNB formation) | 412 nm | High sensitivity, orthogonal validation, endpoint format. | Discontinuous; measures substrate (DTT) depletion, not cofactor turnover directly. |
Visualization: Pathways and Workflows
Diagram Title: MsrB1 Catalytic Cycle and NADPH Consumption Assay Principle
Diagram Title: NADPH Consumption Assay Workflow for Inhibitor Screening
Within the broader thesis research on validating inhibitors of Methionine Sulfoxide Reductase B1 (MsrB1) for therapeutic potential, a robust and quantitative assay for MsrB1 activity is paramount. MsrB1 is a key enzyme in the cellular antioxidant system, specifically reducing methionine-R-sulfoxide residues in proteins. Its activity is coupled to thioredoxin (Trx) and thioredoxin reductase (TrxR), ultimately leading to the oxidation of NADPH. Monitoring NADPH oxidation spectrophotometrically at 340 nm provides a direct, continuous, and sensitive measure of MsrB1 enzymatic activity, forming the cornerstone for inhibitor validation studies.
Assay Principle & Reaction Scheme
The assay is based on the enzymatic recycling system that links MsrB1 activity to NADPH consumption. The principle reaction scheme is as follows:
- MsrB1 reduces its substrate, a methionine-R-sulfoxide (Met-R-SO) residue (often provided as a small peptide like dabsyl-Met-R-SO).
- The oxidized MsrB1 (with a selenenylsulfide bond) is reduced by thioredoxin (Trx), generating oxidized Trx.
- Oxidized Trx is reduced by thioredoxin reductase (TrxR) using NADPH as the electron donor.
- The oxidation of NADPH to NADP⁺ results in a decrease in absorbance at 340 nm, which is monitored in real-time.
The rate of decrease in absorbance at 340 nm (ΔA₃₄₀/min) is directly proportional to MsrB1 activity. In inhibitor validation, test compounds are introduced to the reaction, and a reduction in this rate indicates inhibitory activity.
Table 1: Typical Kinetic Parameters for the MsrB1/NADPH Coupled Assay
| Parameter | Value | Description / Condition |
|---|---|---|
| Extinction Coefficient (ε) of NADPH | 6220 M⁻¹cm⁻¹ | At 340 nm, pH 7.0-7.6 |
| Linear Detection Range | 0.01 – 2.0 ΔA₃₄₀/min | Standard 1 cm pathlength |
| Assay Volume | 50 – 200 µL | Standard 96-well plate format |
| Reaction Temperature | 25°C or 37°C | Controlled by plate reader |
| Typical MsrB1 Km (for substrate) | 5 – 25 µM | Varies with peptide substrate |
| Z'-Factor for HTS | >0.7 | Indicative of a robust assay |
Table 2: Example Inhibitor Screening Data (IC₅₀ Determination)
| Compound ID | IC₅₀ (µM) | 95% Confidence Interval | Hill Slope | Notes |
|---|---|---|---|---|
| DMSO Control | N/A | N/A | N/A | 100% Activity |
| Reference Inhibitor A | 1.5 | 1.2 – 1.9 | -1.1 | Known weak binder |
| Test Compound B | 0.12 | 0.09 – 0.15 | -0.9 | Potent candidate |
| Test Compound C | >50 | N/A | N/A | Inactive |
Detailed Experimental Protocols
Protocol 1: Standard MsrB1 Activity Assay for Inhibitor Screening
Objective: To measure initial velocity of MsrB1 and determine inhibitor IC₅₀ values. Materials: See "The Scientist's Toolkit" below. Procedure:
- Master Mix Preparation (in assay buffer): In a 1.5 mL tube, combine the following for a single 100 µL reaction (prepare a bulk mix for n+2 reactions):
- Assay Buffer (HEPES/KOH, pH 7.4): to volume
- NADPH: 200 µM final concentration
- Thioredoxin (Trx): 10 µM final concentration
- Thioredoxin Reductase (TrxR): 0.5 U/mL final concentration
- Recombinant MsrB1 enzyme: 10-50 nM final concentration (pre-optimized for linear rate)
- Reaction Assembly:
- Aliquot 90 µL of Master Mix into each well of a clear-bottom 96-well plate.
- Add 5 µL of test compound (dissolved in DMSO) or DMSO vehicle control to appropriate wells. Pre-incubate for 10 minutes at assay temperature.
- Initiate the reaction by adding 5 µL of the peptide substrate (dabsyl-Met-R-SO) to a final concentration of 50-200 µM (pre-optimized). Mix gently by pipetting.
- Data Acquisition:
- Immediately place the plate in a pre-warmed (37°C) microplate reader.
- Monitor the decrease in absorbance at 340 nm every 20-30 seconds for 10-15 minutes.
- Data Analysis:
- Plot A₃₄₀ vs. time. Use the linear portion (typically first 3-8 minutes) to calculate the slope (ΔA₃₄₀/min).
- Activity (%) = (Slope with inhibitor / Slope with DMSO control) * 100.
- Fit %Activity vs. log[Inhibitor] to a 4-parameter logistic model to determine IC₅₀.
Protocol 2: Michaelis-Menten Kinetics in Presence of Inhibitor
Objective: To determine the mode of inhibition (competitive, non-competitive, etc.). Procedure:
- Perform Protocol 1 across a range of substrate concentrations (e.g., 5, 10, 25, 50, 100, 200 µM).
- Perform this matrix at several fixed concentrations of the inhibitor (e.g., 0, 0.5xIC₅₀, 1xIC₅₀, 2xIC₅₀).
- Plot initial velocity (v₀) vs. substrate concentration [S] for each inhibitor concentration.
- Fit data to the Michaelis-Menten equation and derive apparent Km and Vmax values.
- Generate double-reciprocal (Lineweaver-Burk) plots to visualize inhibition pattern.
The Scientist's Toolkit
Table 3: Key Research Reagent Solutions
| Item | Function in Assay | Typical Storage/Handling |
|---|---|---|
| Recombinant Human MsrB1 | The target enzyme. Catalyzes the reduction of methionine-R-sulfoxide. | -80°C in aliquots. Avoid freeze-thaw cycles. |
| Thioredoxin (Trx) System | Electron carrier. Directly reduces oxidized MsrB1. | -80°C. Thaw on ice. |
| Thioredoxin Reductase (TrxR) | Regenerates reduced Trx using NADPH. | -80°C. Keep activity-specific. |
| β-NADPH (Tetrasodium Salt) | Primary electron donor. Oxidation measured at 340 nm. | -20°C, desiccated. Prepare fresh solution in assay buffer. |
| Dabsyl-Met-R-SO Peptide | Standardized substrate for MsrB1. | -20°C, lyophilized. Dissolve in DMSO or assay buffer. |
| HEPES/KOH Assay Buffer (pH 7.4) | Maintains physiological pH for optimal enzyme activity. | 4°C. Contains EDTA (1 mM) to chelate metals. |
| Test Inhibitor Compounds | Potential drug candidates to be validated. Dissolved in DMSO. | -20°C. Use at final [DMSO] ≤1% (v/v). |
| Clear 96-Well Microplates | Reaction vessel compatible with plate readers. | Use plates with low UV absorbance. |
Visualizations
NADPH-Coupled MsrB1 Catalytic Cycle (66 chars)
Inhibitor Screening Assay Workflow (44 chars)
Within the broader thesis investigating novel inhibitors of Methionine Sulfoxide Reductase B1 (MsrB1) for therapeutic potential, the quantification of enzymatic activity is paramount. MsrB1, a key enzyme in the repair of oxidative damage to methionine residues, utilizes NADPH as a reducing equivalent. Therefore, a direct and continuous NADPH consumption assay provides a robust, high-throughput method for inhibitor validation. This protocol details the preparation of recombinant MsrB1, kinetic measurement of its activity via NADPH oxidation, and systematic data collection for determining inhibitor potency (IC50/Ki).
Research Reagent Solutions & Essential Materials
| Item | Specification/Composition | Function |
|---|---|---|
| Recombinant Human MsrB1 | ≥95% purity, in 20mM Tris-HCl, pH 7.5, 100mM NaCl, 10% glycerol. | The target enzyme for inhibitor validation studies. |
| NADPH | β-Nicotinamide adenine dinucleotide 2'-phosphate, tetrasodium salt. 100mM stock in assay buffer. | Reducing cofactor; its oxidation is monitored spectrophotometrically at 340 nm. |
| DMSO | Molecular biology grade, ≥99.9%. | Universal solvent for small molecule inhibitor stocks. |
| Methionine-R-Sulfoxide (Met-R-SO) | Substrate for MsrB1. 200mM stock in H₂O. | The specific oxidized methionine substrate reduced by MsrB1. |
| Assay Buffer | 50mM HEPES, pH 7.4, 150mM NaCl, 1mM EDTA, 0.01% Tween-20. | Provides optimal ionic strength, pH, and reduces non-specific binding. |
| Test Inhibitors | Small molecule compounds in DMSO, 10mM stocks. | Potential therapeutic agents for validation. |
| 96-Well UV-Transparent Plate | Polystyrene, flat-bottom. | Vessel for high-throughput kinetic measurements. |
| Microplate Spectrophotometer | Capable of kinetic reads at 340 nm. | Instrument for continuous monitoring of NADPH consumption. |
Protocol 1: Recombinant MsrB1 Sample Preparation
Objective: To prepare an active, purified MsrB1 enzyme stock suitable for kinetic assays. Methodology:
- Thaw & Dilution: Thaw an aliquot of recombinant MsrB1 on ice. Centrifuge briefly to collect contents.
- Desalting/Buffer Exchange: Perform a quick buffer exchange into Assay Buffer using a Zeba Spin Desalting Column (7K MWCO) to remove glycerol and storage salts. Follow manufacturer's instructions.
- Protein Quantification: Determine the final protein concentration using a Bradford or BCA assay against a BSA standard curve.
- Working Solution Preparation: Dilute the exchanged MsrB1 to a 2X working concentration of 100 nM in Assay Buffer. Keep on ice. Note: The final assay concentration is 50 nM.
Protocol 2: Kinetics Measurement & Inhibitor Screening
Objective: To measure the initial velocity of MsrB1-catalyzed NADPH consumption in the presence and absence of inhibitors. Workflow:
Title: NADPH Consumption Assay Workflow for MsrB1 Inhibition.
Detailed Procedure:
- Master Mix (MM): Prepare sufficient MM for all reactions: 80 µM NADPH (from 100mM stock) in Assay Buffer. Protect from light.
- Plate Setup: In a 96-well plate, add 2 µL of inhibitor (at varying concentrations) or DMSO (for controls) to appropriate wells. Include negative controls (No Enzyme) and positive controls (No Inhibitor).
- Add Enzyme & Pre-incubate: Add 98 µL of the MsrB1 Working Solution (100 nM) to all wells except "No Enzyme" controls (add 98 µL Buffer instead). Seal, mix gently, and pre-incubate for 10 minutes at room temperature.
- Reaction Initiation: Using a multi-channel pipette, rapidly add 100 µL of Master Mix containing substrate to achieve final concentrations: 50 nM MsrB1, 40 µM NADPH, 500 µM Met-R-SO, 1% DMSO, and inhibitor (0-100 µM range) in a 200 µL total volume.
- Data Acquisition: Immediately place the plate in a pre-warmed microplate reader. Kinetically measure absorbance at 340 nm every 20 seconds for 5 minutes at 25°C.
Protocol 3: Data Collection & Analysis
Objective: To process raw kinetic data and determine inhibitor potency. Methodology:
- Initial Velocity (V₀) Calculation: For each well, plot Absorbance at 340 nm vs. time. Calculate V₀ as the slope of the linear decrease over the first 2-3 minutes (ΔA340/min). Ensure linear R² > 0.98.
- Normalization: Normalize all V₀ values to the mean of the "No Inhibitor" (100% activity) and "No Enzyme" (0% activity) controls on the same plate.
- Dose-Response & IC50: For each inhibitor, plot normalized activity (%) against log10[Inhibitor]. Fit data to a 4-parameter logistic (sigmoidal) model using software (e.g., GraphPad Prism):
Activity = Bottom + (Top-Bottom) / (1 + 10^((LogIC50 - Log[Inhibitor])*HillSlope)) - Data Reporting: Summarize key quantitative findings.
Table 1: Representative Kinetic Data for Candidate MsrB1 Inhibitor "X"
| Inhibitor [µM] | Mean ΔA340/min (x10⁻³) | SD | Normalized Activity (%) |
|---|---|---|---|
| 0 (DMSO Control) | -8.50 | 0.42 | 100.0 |
| 0.1 | -7.05 | 0.38 | 82.9 |
| 1.0 | -4.10 | 0.31 | 48.2 |
| 10.0 | -1.22 | 0.15 | 14.3 |
| 100.0 | -0.55 | 0.10 | 6.5 |
| No Enzyme | -0.25 | 0.05 | 0.0 |
Table 2: Calculated IC50 Values for Candidate Inhibitors
| Compound ID | IC50 (µM) | 95% CI | Hill Slope | R² of Fit |
|---|---|---|---|---|
| Inhibitor X | 1.05 | (0.89 - 1.24) | -1.1 | 0.998 |
| Inhibitor Y | 15.30 | (12.1 - 19.4) | -0.9 | 0.992 |
| Reference Std. | 0.45 | (0.38 - 0.53) | -1.2 | 0.999 |
Pathway Context: MsrB1 in Redox Metabolism & Inhibition
Title: MsrB1 Reduction Pathway and Site of Inhibitor Action.
This detailed walkthrough provides a standardized framework for preparing MsrB1, conducting NADPH consumption-based kinetics, and collecting robust data for inhibitor validation. The assay's continuous format allows for precise determination of initial velocities, essential for calculating reliable IC50 values. Integration of these protocols into the thesis research enables the quantitative comparison of novel compounds, guiding the selection of lead candidates for further drug development in targeting redox-related pathologies.
This application note details protocols for validating Metallo-Oxidoreductase MsrB1 inhibitors using an NADPH consumption assay, a critical component of research into redox-based therapeutic interventions. The methodology focuses on quantifying enzymatic activity and inhibitor efficacy through precise kinetic data analysis.
Experimental Protocols
NADPH Consumption Assay for MsrB1 Activity
Principle: Recombinant MsrB1 reduces methionine sulfoxide, utilizing thioredoxin as a reductant, which is regenerated by thioredoxin reductase (TR) using NADPH. Inhibitor screening is performed by monitoring the decrease in NADPH absorbance at 340 nm.
Detailed Protocol:
- Reaction Cocktail (100 µL final volume):
- 50 mM HEPES buffer, pH 7.5
- 150 mM NaCl
- 10 µM recombinant human MsrB1
- 10 µM E. coli Thioredoxin (Trx)
- 100 nM E. coli Thioredoxin Reductase (TR)
- 200 µM Methionine-R-Sulfoxide (Met-R-SO) substrate
- 200 µM NADPH
- Candidate inhibitor at desired concentration (e.g., 0.1, 1, 10 µM) in DMSO (final DMSO ≤ 1% v/v).
- Control wells contain DMSO vehicle only.
- Procedure: a. Prepare all reagents and pre-incubate at 30°C. b. In a 96-well quartz microplate, add buffer, NaCl, Trx, TR, and MsrB1. c. Add inhibitor or vehicle and incubate for 10 minutes at 30°C. d. Initiate the reaction by simultaneous addition of Met-R-SO and NADPH using a multichannel pipette. e. Immediately transfer plate to a pre-warmed (30°C) plate reader. f. Record the absorbance at 340 nm every 15 seconds for 10 minutes. g. Perform all measurements in triplicate.
Data Analysis Protocol for Reaction Rate and Inhibition
- Blank Subtraction: Subtract the average absorbance of a no-enzyme control from all sample reads.
- Initial Rate Calculation:
- Plot absorbance at 340 nm (A₃₄₀) vs. time (seconds) for each well.
- Identify the linear phase (typically the first 120-180 seconds).
- Perform linear regression on this interval.
- The slope (ΔA₃₄₀/Δt) is the initial velocity (Vᵢ) in absorbance units per second.
- Reaction Rate Calculation:
- Apply the Beer-Lambert law:
Δ[NADPH]/Δt = - (Vᵢ) / (ε * l) - Where ε (molar extinction coefficient for NADPH at 340 nm) = 6220 M⁻¹cm⁻¹, and l (pathlength) = typically 0.7 cm for a 100 µL well volume.
- Final reaction rate is expressed as µM NADPH consumed per second (µM/s).
- Apply the Beer-Lambert law:
- Percent Inhibition Calculation:
% Inhibition = [1 - (Rate_inhibited / Rate_control)] * 100- Where
Rate_controlis the average reaction rate for the DMSO vehicle control.
Data Presentation
Table 1: Calculated Reaction Rates and Percent Inhibition for Candidate Compounds
| Compound ID | Mean Reaction Rate (µM/s) ± SD | % Inhibition (at 10 µM) | IC₅₀ (µM) [if determined] |
|---|---|---|---|
| DMSO Control | 0.152 ± 0.008 | 0.0 | N/A |
| Cmpd-A001 | 0.021 ± 0.003 | 86.2 ± 2.1 | 0.45 ± 0.07 |
| Cmpd-A002 | 0.098 ± 0.007 | 35.5 ± 4.6 | >10 |
| Cmpd-B107 | 0.005 ± 0.001 | 96.7 ± 0.7 | 0.12 ± 0.02 |
| Cmpd-C225 | 0.145 ± 0.009 | 4.6 ± 5.9 | N/D |
SD: Standard Deviation (n=3); N/D: Not Determined.
Table 2: Key Research Reagent Solutions
| Reagent / Material | Function / Rationale |
|---|---|
| Recombinant hMsrB1 | Target enzyme; catalyzes the reduction of methionine sulfoxide. |
| Met-R-Sulfoxide | Specific substrate for the MsrB1 isoform. |
| NADPH | Reducing cofactor; its consumption is the direct spectrophotometric readout. |
| Thioredoxin (Trx) System (Trx + TR) | Electron transfer chain that shuttles reducing equivalents from NADPH to MsrB1. |
| HEPES Buffer (pH 7.5) | Maintains physiological pH for optimal enzyme activity. |
| Quartz Microplate | Allows accurate UV absorbance readings at 340 nm. |
Visualizations
Title: MsrB1 Redox Pathway and Inhibitor Site
Title: NADPH Assay and Data Analysis Workflow
Solving Common Problems and Enhancing Assay Performance and Sensitivity
Within the broader thesis on validating novel inhibitors of Methionine Sulfoxide Reductase B1 (MsrB1), the NADPH consumption assay is a critical biochemical high-throughput screening (HTS) tool. The assay monitors the oxidation of NADPH to NADP⁺, catalyzed by thioredoxin reductase, as it regenerates reduced thioredoxin, the electron donor for MsrB1. A robust signal-to-background ratio is paramount for accurately quantifying inhibitor potency (IC₅₀). This document details systematic troubleshooting for the prevalent issues of low signal (poor dynamic range) and high background, focusing on reagent stability and contamination.
Key Reagent Stability Profiles & Quantitative Data
The core assay components are susceptible to degradation, directly impacting signal magnitude and background noise. Current literature and vendor data sheets inform the following stability parameters.
Table 1: Stability Profile of Core NADPH-MsrB1 Assay Reagents
| Reagent | Primary Stability Concern | Recommended Handling & Storage | Observed Signal Impact (if compromised) | Typical Half-life (under assay conditions) |
|---|---|---|---|---|
| NADPH (Tetrasodium Salt) | Oxidation by ambient O₂, photodegradation. | Prepare fresh in cold, degassed buffer daily; aliquot & store at -80°C, desiccated; protect from light. | High Background: Pre-oxidized NADPH increases initial A₃₄₀. Low Signal: Decreased maximum consumption rate. | ~2-4 hours in assay buffer at 25°C. |
| Dithiothreitol (DTT) | Oxidation to disulfide in stock solutions. | Use high-purity grade; prepare fresh 1M stock in water weekly; store at -20°C under inert gas. | Low Signal: Inefficient reduction of Trx/TrxR system. | ~1 week as 1M stock at -20°C. |
| E. coli Thioredoxin (Trx) | Disulfide formation and aggregation. | Aliquot in working buffer + 10% glycerol; avoid freeze-thaw >5 cycles; store at -80°C. | Low Signal: Reduced electron transfer to MsrB1. | Stable for months at -80°C. |
| E. coli Thioredoxin Reductase (TrxR) | Loss of FAD cofactor, sulfhydryl oxidation. | Aliquot in storage buffer; store at -80°C; keep on ice during use. | Low Signal & High Background: Impaired NADPH oxidation, may cause non-linear kinetics. | Stable for months at -80°C; labile >4 hours on ice. |
| Recombinant Human MsrB1 | Oxidation of active site cysteines. | Store in buffer with 1-5 mM DTT or TCEP; aliquot at high concentration; store at -80°C. | Low Signal: Reduced substrate turnover. | Activity loss ~5%/month at -80°C without reductant. |
| Assay Buffer (Tris/Hepes, pH 7.5-8.0) | Microbial growth, pH drift. | Filter sterilize (0.22 µm), store at 4°C for ≤2 weeks. | High Background: Microbial NADPH oxidases increase background consumption. | 2 weeks at 4°C. |
Detailed Troubleshooting Protocols
Protocol A: Diagnosing the Source of High Background
Objective: Isolate which component contributes to elevated NADPH oxidation in the absence of the target enzyme reaction. Workflow:
- Prepare all reagents fresh or from validated stable aliquots. Use NADPH-free buffer as blank.
- In a 96-well plate, set up the following mixtures in triplicate (100 µL final volume):
- Well 1: Buffer + NADPH.
- Well 2: Buffer + NADPH + TrxR.
- Well 3: Buffer + NADPH + TrxR + Trx.
- Well 4: Buffer + NADPH + TrxR + Trx + DTT.
- Well 5 (Full Background Control): Buffer + NADPH + TrxR + Trx + DTT + Inactivated MsrB1 (heat-treated at 95°C for 10 min).
- Initiate reaction by adding NADPH last. Immediately monitor A₃₄₀ every minute for 15-30 minutes at 25°C.
- Interpretation: A significant slope in Well 1 indicates contaminated NADPH or buffer. An increase in slope from Well 2 to Well 3/4 indicates contaminated or impure TrxR/Trx preparations. The slope of Well 5 is your assay-specific background to subtract.
Protocol B: Verifying Reagent Activity & Assay Dynamic Range
Objective: Confirm each component is functional and the assay yields a robust signal window. Workflow:
- Positive Control (Full Reaction): 50 mM HEPES (pH 7.6), 150 mM NaCl, 1 mM EDTA, 200 µM NADPH, 5 mM DTT, 100 nM TrxR, 10 µM Trx, 50-100 nM active MsrB1, 1-5 mM substrate (e.g., Dabsyl-MetSO). Monitor ΔA₃₄₀/min.
- Negative Control (No Enzyme): As above, replace MsrB1 with storage buffer.
- Component Omission Controls: Run separate reactions omitting DTT, Trx, or TrxR individually from the full reaction.
- Calculate Dynamic Range: (RatePositive Control – RateNegative Control) / Rate_Negative Control. A ratio of <5 indicates poor signal window, necessitating troubleshooting via Table 1.
Visualization of Pathways and Workflows
Title: MsrB1 NADPH Consumption Assay Electron Transfer Pathway
Title: Troubleshooting Decision Tree for Low Signal/High Background
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Robust NADPH-Consumption MsrB1 Assays
| Item | Function in the Assay | Critical Notes for Stability/Contamination |
|---|---|---|
| Ultra-Pure NADPH (Tetrasodium Salt) | Primary spectrophotometric substrate; its oxidation is the measured signal. | Most common culprit. Source from reputable vendors; check absorbance ratio A₂₆₀/A₃₄₀ (<2.3 indicates purity). |
| Lyophilized Trx/TrxR (E. coli) | Regenerable electron transfer cascade linking NADPH oxidation to MsrB1 reduction. | Reconstitute with provided buffer; aliquot immediately to minimize freeze-thaw cycles of working stocks. |
| High-Purity DTT or TCEP | Maintains reducing environment, keeps Trx and MsrB1 cysteines reduced. | TCEP is more stable at neutral pH and preferred for long incubations. Aliquot stock under argon. |
| Absorption-Matched 96-Well Plates | For kinetic readout of NADPH at 340 nm. | Use plates with minimal meniscus and high optical clarity. Ensure compatibility with plate reader. |
| Black, Sealing Foils | Prevents evaporation and atmospheric oxygen ingress during kinetic reads. | Essential for long runs (>30 min). |
| PCR-Tube Style Microcentrifuge Tubes | For storing small, single-use aliquots of critical reagents (NADPH, MsrB1). | Minimizes headspace, reducing oxidation during storage. |
| Filtered, Degassed Assay Buffer | Reaction milieu. | Degassing (via vacuum or sonication) reduces dissolved O₂, slowing NADPH auto-oxidation. |
Thesis Context: NADPH Consumption Assay for MsrB1 Inhibitor Validation Within the broader thesis research on targeting Methionine Sulfoxide Reductase B1 (MsrB1) for therapeutic intervention, the development of a robust, high-throughput screening (HTS) assay is critical. MsrB1 reduces methionine-R-sulfoxide in proteins, utilizing thioredoxin (Trx) as a reductant. In a coupled enzymatic system, Trx reductase (TrxR) regenerates reduced Trx using NADPH, resulting in a detectable decrease in NADPH absorbance at 340 nm. Inhibitors of MsrB1 will attenuate NADPH consumption. The Z'-factor is a key statistical parameter used to assess the quality and robustness of an HTS assay, with values ≥0.5 considered excellent for screening. This protocol details the optimization of enzyme concentrations and reaction conditions to achieve a robust Z'-factor for reliable MsrB1 inhibitor validation.
1. Key Research Reagent Solutions
| Reagent | Function in Assay | Typical Supplier/Note |
|---|---|---|
| Recombinant Human MsrB1 | Target enzyme; catalyzes the reduction of methionine-R-sulfoxide substrate. | Purified in-house or commercial (e.g., R&D Systems). |
| Thioredoxin (Trx) | Immediate electron donor to MsrB1. | Human recombinant. |
| Thioredoxin Reductase (TrxR) | Regenerates reduced Trx, consuming NADPH. | Human recombinant, NADPH-dependent. |
| NADPH | Cofactor; consumption monitored at 340 nm (ε=6220 M⁻¹cm⁻¹). | Lithium salt, high purity. |
| Methionine-R-Sulfoxide (Met-R-SO) | Substrate for MsrB1. | Synthetic, ≥95% purity. |
| Assay Buffer (e.g., Tris, HEPES) | Maintains optimal pH and ionic strength. | Contains EDTA (1 mM) to chelate metals. |
| DMSO (Dimethyl Sulfoxide) | Vehicle for compound/library dissolution. | Low UV absorbance, anhydrous. |
| Putative Inhibitor Compounds | Small molecules for MsrB1 validation. | Dissolved in DMSO, stored at -20°C. |
2. Protocol: Enzyme Titration for Signal Window Optimization Objective: Determine the optimal concentrations of MsrB1 and Trx/TrxR that yield a maximal signal window (difference between positive and negative controls) with minimal variability.
2.1 Materials
- Assay Buffer: 50 mM HEPES, pH 7.5, 1 mM EDTA.
- Stock Solutions: 100 µM MsrB1, 100 µM Trx, 10 U/mL TrxR, 10 mM NADPH, 100 mM Met-R-SO, 10% DMSO (v/v).
- Equipment: 96-well or 384-well clear flat-bottom microplate, multichannel pipettes, plate reader capable of kinetic reads at 340 nm.
2.2 Procedure
- Prepare a 2X master mix of Trx (final concentration range: 0.5-10 µM), TrxR (final range: 0.01-0.5 U/mL), and NADPH (final: 200 µM) in assay buffer.
- Prepare serial dilutions of MsrB1 in assay buffer to yield 2X stocks covering a final concentration range of 5-100 nM.
- In the microplate, add 25 µL of the 2X MsrB1 dilution or assay buffer (for negative control) to designated wells.
- Initiate the reaction by adding 25 µL of the 2X master mix containing Trx/TrxR/NADPH and the substrate Met-R-SO (final concentration: 500 µM).
- Immediately place the plate in the reader and record the decrease in absorbance at 340 nm every 20 seconds for 10 minutes at 25°C.
- Calculate the initial linear reaction rate (ΔmOD₃₄₀/min) for each well.
2.3 Data Analysis Plot reaction rate vs. MsrB1 concentration for each Trx/TrxR condition. The optimal condition is the lowest enzyme concentration that gives a near-maximal rate, ensuring reagent conservation and linear kinetics. Calculate the Signal-to-Noise (S/N) and Signal-to-Background (S/B) ratios. Table 1: Example Optimization Data for Signal Window
| [MsrB1] (nM) | [Trx] (µM) | Rate (ΔmOD/min) Positive Control | Rate (ΔmOD/min) Negative Control (No MsrB1) | S/B Ratio | Coefficient of Variation (CV%) Positive |
|---|---|---|---|---|---|
| 10 | 2.0 | 12.5 ± 0.8 | 0.9 ± 0.1 | 13.9 | 6.4 |
| 20 | 2.0 | 22.1 ± 1.5 | 0.9 ± 0.1 | 24.6 | 6.8 |
| 50 | 2.0 | 25.3 ± 2.1 | 1.0 ± 0.2 | 25.3 | 8.3 |
| 20 | 5.0 | 23.8 ± 1.2 | 1.2 ± 0.1 | 19.8 | 5.0 |
3. Protocol: Z'-Factor Determination under Optimized Conditions Objective: Evaluate the statistical robustness of the finalized assay format.
3.1 Procedure
- Using optimized concentrations from Section 2 (e.g., 20 nM MsrB1, 2 µM Trx, 0.1 U/mL TrxR, 200 µM NADPH, 500 µM Met-R-SO), prepare positive control (full reaction) and negative control (no MsrB1) solutions in bulk.
- Dispense 50 µL of each control solution into 32 wells each of a 384-well plate (n=32).
- Run the kinetic assay as in Section 2.2.
- Calculate the mean (µ) and standard deviation (σ) of the initial rate for both control sets.
3.2 Calculation Calculate the Z'-factor using the formula: Z' = 1 - [ (3σpositive + 3σnegative) / |µpositive - µnegative| ] Table 2: Z'-Factor Calculation from Control Data
| Parameter | Positive Control (n=32) | Negative Control (n=32) |
|---|---|---|
| Mean Rate (ΔmOD/min) | 21.85 | 0.92 |
| Standard Deviation | 1.42 | 0.09 |
| Z'-Factor | 0.78 |
A Z' of 0.78 indicates an excellent assay robust enough for HTS.
4. Protocol: MsrB1 Inhibitor Validation Run Objective: Confirm assay performance by testing putative inhibitors.
4.1 Procedure
- Dispense 0.5 µL of test compound (in DMSO) or DMSO alone (for controls) into assay plates. Final DMSO concentration should be ≤1%.
- Add 24.5 µL of optimized 2X MsrB1 solution to all wells.
- Incubate plate for 10 minutes at 25°C.
- Initiate reaction by adding 25 µL of optimized 2X substrate/cofactor mix (Trx, TrxR, NADPH, Met-R-SO).
- Read kinetically at 340 nm.
- Calculate % Inhibition: [1 - (Ratecompound / Ratepositive_control)] * 100.
Diagrams
Title: MsrB1 Assay Optimization and Z' Assessment Workflow
Title: NADPH-Coupled MsrB1 Activity and Inhibition Pathway
1. Introduction Within the context of NADPH consumption assay development for Methionine Sulfoxide Reductase B1 (MsrB1) inhibitor validation, a major technical hurdle is compound interference. Potential small-molecule inhibitors can directly interfere with assay signal generation through intrinsic fluorescence, absorbance at the detection wavelength, or inherent redox activity, leading to false positives or negatives. These Application Notes detail protocols to identify, quantify, and correct for such interference to ensure robust hit validation.
2. Quantifying and Correcting for Interference The following protocols are designed to be executed prior to primary high-throughput screening (HTS) or during secondary validation of hits from an NADPH consumption assay.
Protocol 2.1: Fluorescence Interference Test Objective: Determine if test compounds exhibit fluorescence at the excitation/emission wavelengths used for resorufin detection (often ~571/585 nm) in coupled enzyme assays. Materials:
- Test compounds (in DMSO, final assay concentration, e.g., 10 µM)
- Assay buffer (e.g., 50 mM Tris-HCl, pH 7.5, 1 mM EDTA)
- Black, clear-bottom 96- or 384-well plates
- Plate reader capable of fluorescence detection Procedure:
- Prepare compound solutions in assay buffer with the same final DMSO concentration as the main assay (e.g., 1%).
- Dispense 100 µL of each compound solution or buffer-only control into plate wells in triplicate.
- Measure fluorescence using the same settings as the primary assay (e.g., Ex 571 nm, Em 585 nm, cutoff 590 nm).
- Calculate fluorescence as a percentage of the background (buffer) signal. A signal >115% of background suggests significant interference.
Protocol 2.2: Absorbance Interference Test Objective: Determine if test compounds absorb light at the critical assay wavelengths (340 nm for NADPH, ~571 nm for resorufin). Materials:
- Test compounds (in DMSO, final assay concentration)
- Assay buffer
- Clear 96- or 384-well plates
- Plate reader capable of absorbance measurement Procedure:
- Prepare compound solutions as in Protocol 2.1.
- Dispense 100 µL into plate wells in triplicate.
- Measure absorbance at 340 nm and 571 nm.
- Calculate the absorbance relative to buffer. An absorbance >0.1 AU above background at either wavelength can cause significant signal artifact.
Protocol 2.3: Redox Interference Test (Chemical Reduction of Probe) Objective: Determine if compounds directly reduce the detection probe (e.g., resazurin) non-enzymatically. Materials:
- Test compounds
- Assay buffer
- Resazurin solution (final concentration matching main assay, e.g., 50 µM)
- Stop solution (e.g., 1% SDS)
- Clear or black plates (depending on detection mode) Procedure:
- In plate wells, mix compound (final concentration), resazurin, and buffer. Omit the enzyme (MsrB1) and enzymatic substrate (Met-O).
- Incubate under assay conditions (e.g., 30 min, 37°C).
- Stop the reaction with SDS.
- Measure fluorescence (Ex/Em 571/585 nm) or absorbance (571 nm).
- A significant increase in resorufin signal compared to a no-compound control indicates direct chemical reduction of the probe.
3. Data Presentation: Summary of Interference Thresholds
Table 1: Thresholds for Flagging Compound Interference
| Interference Type | Assay Readout Affected | Test Protocol | Threshold for Concern | Corrective Action |
|---|---|---|---|---|
| Fluorescence | Fluorescence increase (Resorufin) | 2.1 | Signal >115% of buffer control | Use orthogonal assay (e.g., absorbance). Apply correction factor if consistent. |
| Absorbance at 340 nm | Absorbance decrease (NADPH) | 2.2 | A340 >0.1 AU above buffer | Switch to fluorescence-coupled detection. Pre-screen and exclude. |
| Absorbance at 571 nm | Absorbance/Fl. increase (Resorufin) | 2.2 | A571 >0.1 AU above buffer | Use kinetic read, subtract initial absorbance. Pre-screen and exclude. |
| Redox Activity | False positive signal increase | 2.3 | Signal >3 SDs above negative control | Confirm hits in an orthogonal, non-redox assay (e.g., HPLC, NADPH absorbance kinetics). |
Table 2: Example Interference Data for Candidate MsrB1 Inhibitors
| Compound ID | Primary Assay (% Inhibition) | Fluorescence (% of Control) | A340 (AU) | A571 (AU) | Redox Activity (RFU) | Validated Hit? |
|---|---|---|---|---|---|---|
| Cmpd A | 85% | 102% | 0.05 | 0.02 | 105 | Yes |
| Cmpd B | 92% | 215% | 0.01 | 0.01 | 110 | No (Fluor. Interf.) |
| Cmpd C | 78% | 98% | 0.45 | 0.03 | 95 | No (A340 Interf.) |
| Cmpd D | 95% | 105% | 0.04 | 0.40 | 650 | No (Redox Interf.) |
RFU: Relative Fluorescence Units; AU: Absorbance Units. Control RFU = 100.
4. Integrated Workflow for Inhibitor Validation
Title: Workflow for MsrB1 Inhibitor Validation with Interference Triage
5. The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Interference Testing in NADPH Assays
| Item | Function/Benefit | Example/Notes |
|---|---|---|
| Recombinant MsrB1 Enzyme | Target enzyme for inhibitor validation. Essential for orthogonal activity assays. | Purified human or mouse MsrB1; confirm activity via DTNB or HPLC assays. |
| NADPH (Tetrasodium Salt) | Essential redox cofactor for the primary consumption assay. | High-purity, store at -80°C in aliquots. Monitor A340 for stability. |
| Resazurin Sodium Salt | Redox-sensitive probe for coupled fluorescence detection. | Used in redox interference test. Prepare fresh or store aliquots protected from light. |
| Dithiothreitol (DTT) | Physiological reducing agent for Msr enzyme activity. | Can interfere with some redox probes; use consistent concentrations. |
| L-Methionine (R)-Sulfoxide (Met-R-O) | Substrate for MsrB1. Critical for confirmatory orthogonal assays. | Ensure stereochemical purity (R-form for MsrB1). |
| Black/Clear-Bottom 384-Well Plates | Optimal plates for fluorescence and absorbance readings in low volumes. | Essential for running parallel interference tests. |
| DTNB (Ellman's Reagent) | For orthogonal, non-optical Msr activity assay. Measures free thiol production. | Colorimetric readout at 412 nm; subject to absorbance interference. |
| Microplate Reader | Capable of kinetic UV-Vis and fluorescence measurements. | Requires temperature control for kinetic NADPH measurements at 340 nm. |
Application Notes
Robust assay validation is the cornerstone of reliable high-throughput screening (HTS) and drug discovery. Within the context of a broader thesis on NADPH consumption assay for Methionine Sulfoxide Reductase B1 (MsrB1) inhibitor validation, this document outlines critical validation parameters. MsrB1, a key enzyme in antioxidant defense and redox regulation, is a promising therapeutic target. The coupled enzyme assay, which monitors NADPH consumption spectrophotometrically, requires stringent validation to ensure data integrity for inhibitor characterization. The core pillars of this validation are precision (repeatability and intermediate precision), reproducibility (across days, analysts, and instruments), and DMSO tolerance, as inhibitors are typically solubilized in DMSO.
1. Precision & Reproducibility: For enzymatic assays, precision is measured primarily through the coefficient of variation (%CV) of replicate measurements. Intra-assay precision (repeatability) assesses variability within a single plate or run. Inter-assay precision (intermediate precision) evaluates variability across different days, analysts, or plate readers. Acceptance criteria for HTS-ready assays typically require a Z'-factor ≥ 0.5, signal-to-background (S/B) ratio ≥ 3, and %CV for positive and negative controls < 10-15%. In the MsrB1 NADPH consumption assay, this involves testing multiple replicates of high-activity (no inhibitor) and low-activity (full enzymatic inhibition control) samples across validation runs.
2. DMSO Tolerance: As candidate inhibitors are stored in DMSO stocks, the assay's functional tolerance to final DMSO concentrations is non-negotiable. DMSO can affect enzyme kinetics, protein stability, and signal detection. A systematic tolerance test determines the maximum DMSO concentration that does not statistically alter the assay signal, typically aiming for ≤1% final concentration. The validated DMSO level then becomes the standard for all subsequent screening and IC50 determinations.
Key Validation Data Summary
Table 1: Summary of Precision Validation Parameters for an MsrB1 NADPH Consumption Assay
| Parameter | Measurement | Target Acceptance Criteria | Example Result |
|---|---|---|---|
| Signal-to-Background (S/B) | Mean Signal (No Inhibitor) / Mean Background (No Enzyme) | ≥ 3 | 8.5 |
| Signal Window | 1 - (3SD_high + 3SDlow)/|Meanhigh - Mean_low| | N/A for Z' calculation | 0.75 |
| Z'-Factor | 1 - (3SD_high + 3SDlow)/|Meanhigh - Mean_low| | ≥ 0.5 | 0.78 |
| Intra-assay Precision (%CV) | CV of High & Low Controls (within plate, n=16) | < 10% | High: 4.2%; Low: 5.1% |
| Inter-assay Precision (%CV) | CV of High & Low Controls (across 3 days, 2 analysts) | < 15% | High: 8.7%; Low: 9.3% |
Table 2: DMSO Tolerance Test for MsrB1 Activity
| Final DMSO Concentration (% v/v) | Relative MsrB1 Activity (% of 0% DMSO Control) | Observation |
|---|---|---|
| 0.0 | 100.0 ± 3.5 | Baseline activity. |
| 0.5 | 99.1 ± 4.2 | No significant inhibition. |
| 1.0 | 97.5 ± 5.0 | No significant inhibition. |
| 1.5 | 92.1 ± 6.8 | Mild inhibition observed. |
| 2.0 | 85.4 ± 7.3 | Significant inhibition (p<0.05). |
| Validated Max Concentration | 1.0% | Recommended for all screening. |
Experimental Protocols
Protocol 1: Intra- and Inter-Assay Precision Validation for MsrB1 NADPH Consumption
Objective: To determine the repeatability (intra-assay) and intermediate precision (inter-assay) of the MsrB1 enzymatic activity measurement.
Materials: Purified recombinant human MsrB1 enzyme, NADPH, DTT, Methionine-R-sulfoxide (Met-R-SO) substrate, assay buffer (e.g., 50 mM Tris-HCl, pH 7.5, 150 mM NaCl), clear 96-well or 384-well microplates, plate reader capable of reading absorbance at 340 nm.
Procedure:
- Solution Preparation: Prepare fresh assay buffer. Prepare a master mix containing NADPH (final conc. 100-200 µM) and DTT (final conc. 1-2 mM) in buffer. Prepare Met-R-SO substrate stock in buffer (final conc. 0.5-1 mM). Dilute MsrB1 enzyme in buffer to 2x the desired final concentration.
- Plate Layout: Designate columns/wells for High Control (enzyme + substrate), Low Control (enzyme + no substrate, or buffer only), and Background (substrate + no enzyme). Use a minimum of n=16 replicates per control for intra-assay precision.
- Assay Assembly (for High Control): Add 25 µL of 2x enzyme solution to each well. Add 25 µL of substrate/master mix solution. For Low Control, add 25 µL of 2x enzyme to 25 µL of buffer/master mix without substrate.
- Kinetic Measurement: Immediately place plate in pre-warmed (37°C) plate reader. Monitor the decrease in absorbance at 340 nm (A340) every 20-30 seconds for 10-15 minutes.
- Data Analysis: Calculate the linear rate (ΔA340/min) for each well. Calculate the mean, standard deviation (SD), and %CV for each control group. Compute Z'-factor.
- Inter-assay Precision: Repeat the entire protocol on three separate days, using two different analysts and/or different plate readers if possible. Pool the control data from all runs to calculate the overall inter-assay %CV.
Protocol 2: Determination of DMSO Tolerance
Objective: To establish the maximum concentration of DMSO that does not interfere with MsrB1 enzymatic activity.
Materials: As in Protocol 1, plus anhydrous DMSO (high-grade, sterile).
Procedure:
- DMSO Series Preparation: Prepare a dilution series of DMSO in assay buffer to create 2x solutions that will yield final well concentrations of 0%, 0.5%, 1.0%, 1.5%, 2.0%, and 3.0% v/v upon a 1:1 dilution with other components.
- Assay Assembly with DMSO: For each DMSO concentration, set up High Control wells. Mix 25 µL of 2x enzyme solution with 25 µL of the 2x substrate/master mix solution prepared with the corresponding 2x DMSO buffer. Use n=8 replicates per condition.
- Control Wells: Include a 0% DMSO High Control (standard buffer) as the reference for 100% activity.
- Kinetic Measurement: Perform kinetic reading as in Protocol 1.
- Data Analysis: Calculate the initial reaction rate (ΔA340/min) for each well. Normalize the mean rate of each DMSO condition to the mean rate of the 0% DMSO control. Perform a one-way ANOVA with Dunnett's post-hoc test to identify the lowest DMSO concentration causing statistically significant (p < 0.05) inhibition. The highest concentration with no significant inhibition is the validated tolerance limit.
Mandatory Visualization
Title: Three Pillars of Assay Validation
Title: MsrB1 Redox Cycle & NADPH Consumption Assay
Title: Assay Validation Workflow for MsrB1 Screening
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for MsrB1 NADPH Consumption Assay Validation
| Item | Function / Role in Validation |
|---|---|
| Recombinant Human MsrB1 Protein | The purified target enzyme. Essential for establishing baseline activity, precision, and inhibitor sensitivity. Batch-to-batch consistency is critical for reproducibility. |
| β-NADPH (Tetrasodium Salt) | The spectroscopic probe. Its oxidation (A340 decrease) is directly coupled to MsrB1 activity via the thioredoxin system. High purity ensures consistent initial absorbance. |
| Methionine-R-sulfoxide (Met-R-SO) | The enzyme-specific substrate. Validates correct enzymatic function and provides the signal window for precision calculations. |
| Dithiothreitol (DTT) / Thioredoxin System | Provides reducing equivalents. DTT can be used directly or as part of a coupled Thioredoxin/Thioredoxin Reductase system to recycle oxidized MsrB1. |
| Anhydrous DMSO (High-Grade) | Universal solvent for small molecule inhibitors. Used for tolerance testing and is the vehicle for all test compounds. Low water content is vital for stock solution stability. |
| Clear Flat-Bottom Microplates (96/384-well) | Assay vessel. Must have low UV absorbance and be compatible with the plate reader. Consistency in well geometry is key for reproducible pathlength and signal. |
| UV-Vis Plate Reader | Detection instrument. Must have stable temperature control (37°C) and precise kinetic measurement capability at 340 nm. Calibration is part of inter-assay reproducibility. |
| Assay Buffer Components (Tris, NaCl) | Maintains optimal pH and ionic strength for enzyme activity. Consistent preparation is crucial for day-to-day reproducibility. |
Confirming Specificity: Orthogonal Validation and Comparative Method Analysis
Correlating NADPH Consumption with Direct Activity Assays (e.g., HPLC-Based Substrate Conversion).
Application Notes
Within the context of a thesis on MsrB1 inhibitor validation, integrating NADPH consumption assays with direct substrate conversion analysis is critical for robust, orthogonal enzyme inhibition characterization. NADPH consumption provides a continuous, indirect measure of methionine sulfoxide reductase activity via the thioredoxin/thioredoxin reductase recycling system. However, to rule out assay interference and confirm direct target engagement, this kinetic data must be correlated with a direct, end-point measurement of the primary enzymatic function: the reduction of methionine-R-sulfoxide (Met-R-SO) to methionine.
These Application Notes detail the parallel execution of a coupled spectrophotometric NADPH consumption assay and an HPLC-based Met-R-SO conversion assay, establishing a validated framework for confirming MsrB1 inhibitor potency and mechanism.
Quantitative Data Correlation Table
Table 1: Correlation of IC₅₀ Values from NADPH Consumption and HPLC-Based Assays for Candidate MsrB1 Inhibitors.
| Compound ID | NADPH Consumption Assay IC₅₀ (µM) [Mean ± SD, n=3] | HPLC-Based Substrate Conversion Assay IC₅₀ (µM) [Mean ± SD, n=3] | Correlation Ratio (HPLC/NADPH) | Notes |
|---|---|---|---|---|
| INH-001 | 1.52 ± 0.21 | 1.78 ± 0.31 | 1.17 | Strong correlation suggests direct inhibition. |
| INH-002 | 0.87 ± 0.11 | 15.40 ± 2.50 | 17.70 | High disparity suggests potential assay interference in NADPH assay. |
| DMSO Control | N/A | N/A | N/A | 100% activity baseline. |
| Selenite (Positive Ctrl) | 0.05 ± 0.01 | 0.08 ± 0.02 | 1.60 | Known direct inhibitor; validates both assays. |
Experimental Protocols
Protocol 1: Coupled Spectrophotometric NADPH Consumption Assay for MsrB1 Objective: To continuously measure MsrB1 activity via the depletion of NADPH absorbance at 340 nm. Reagents: Recombinant human MsrB1, Thioredoxin (Trx1), Thioredoxin Reductase (TrxR1), NADPH, Dithiothreitol (DTT), Methionine-R-Sulfoxide (Met-R-SO), Assay Buffer (50 mM Tris-HCl, 150 mM NaCl, 1 mM EDTA, pH 7.5). Procedure:
- Prepare a master mix containing assay buffer, 200 µM NADPH, 10 µM Trx1, 0.5 µM TrxR1, and 1 mM DTT.
- Aliquot 90 µL of master mix into a 96-well quartz microplate. Add 5 µL of inhibitor solution or DMSO control.
- Initiate the reaction by adding 5 µL of MsrB1 enzyme (final concentration 100 nM) and 5 µL of Met-R-SO substrate (final concentration 500 µM). Final reaction volume: 105 µL.
- Immediately monitor the decrease in absorbance at 340 nm (ε₃₄₀ = 6220 M⁻¹cm⁻¹) for 10 minutes at 25°C using a plate reader.
- Calculate initial reaction velocities (Vo) from the linear slope. Determine IC₅₀ values by fitting Vo vs. inhibitor concentration to a four-parameter logistic model.
Protocol 2: HPLC-Based Direct Substrate Conversion Assay for MsrB1 Objective: To directly quantify the conversion of Met-R-SO to methionine as a validated endpoint measure of MsrB1 activity. Reagents: Recombinant human MsrB1, Methionine-R-Sulfoxide (Met-R-SO), Dithiothreitol (DTT), Methionine standard, Derivatization Reagent: AccQ•Fluor Reagent Kit (Waters), HPLC-grade solvents. Procedure:
- In a 1.5 mL tube, combine assay buffer, 2 mM DTT, 200 µM Met-R-SO, and inhibitor or DMSO.
- Start the reaction by adding MsrB1 (final 200 nM). Final volume: 50 µL. Incubate at 37°C for 30 minutes.
- Terminate the reaction by adding 10 µL of 20% (v/v) trichloroacetic acid and vortex. Place on ice for 10 min, then centrifuge at 16,000 × g for 10 min to pellet protein.
- Derivatize the supernatant: Mix 10 µL of clear supernatant with 70 µL of borate buffer and 20 µL of AccQ•Fluor reagent. Incubate at 55°C for 10 min.
- HPLC Analysis: Inject derivatized samples onto a reversed-phase C18 column. Use a gradient of 10 mM NaOAc (pH 5.0) and acetonitrile. Detect methionine and Met-R-SO derivatives by fluorescence (Ex 250 nm, Em 395 nm).
- Quantify peak areas. Calculate % conversion to methionine. Determine IC₅₀ by fitting % conversion vs. inhibitor concentration to a four-parameter logistic model.
Visualization
Diagram 1: MsrB1 catalytic & NADPH coupling pathway
Diagram 2: Orthogonal assay workflow for inhibitor validation
The Scientist's Toolkit
Table 2: Essential Research Reagent Solutions for MsrB1 Activity/Inhibition Profiling.
| Item | Function & Relevance in Assays |
|---|---|
| Recombinant Human MsrB1 | Catalytic enzyme of interest; source of activity for both assay formats. Must be highly active and pure. |
| Thioredoxin (Trx1)/Thioredoxin Reductase (TrxR1) | Enzymatic recycling system for oxidized MsrB1. Essential components of the coupled NADPH consumption assay. |
| NADPH (Tetrasodium Salt) | Reducing cofactor. Its oxidation is monitored at 340 nm to infer MsrB1 activity indirectly. |
| Methionine-R-Sulfoxide (Met-R-SO) | Physiological substrate for MsrB1. Converted to methionine in the direct activity assay. |
| AccQ•Fluor Reagent Kit | Derivatization kit for primary amines. Enables sensitive fluorescence detection of methionine and Met-R-SO by HPLC. |
| Dithiothreitol (DTT) | Maintaining reducing conditions and providing electrons in the direct HPLC assay where the Trx system is absent. |
| Selenite (Sodium Selenite) | Known potent, direct MsrB1 inhibitor. Serves as a critical positive control for inhibition in both assays. |
| Quartz 96-Well Microplate | Essential for accurate UV absorbance measurements at 340 nm in the NADPH consumption assay. |
This application note details a comprehensive cellular validation strategy for MsrB1 inhibitors, utilizing an NADPH consumption assay as a core pharmacodynamic readout. Framed within a broader thesis on methionine sulfoxide reductase B1 (MsrB1) as a therapeutic target, this guide provides protocols to directly link biochemical inhibition to cellular target engagement and functional efficacy, enabling confident progression of lead compounds in drug discovery pipelines.
Methionine sulfoxide reductase B1 (MsrB1) is a key enzyme in redox homeostasis, reducing methionine-R-sulfoxide in proteins. Its activity is NADPH-dependent. In a cellular context, inhibition of MsrB1 leads to an accumulation of oxidized methionine residues and a measurable decrease in the rate of NADPH consumption. Monitoring cellular NADPH levels thus serves as a direct, quantitative indicator of MsrB1 target engagement and functional inhibition. This approach bridges the gap between in vitro enzyme assays and complex phenotypic outcomes.
Key Research Reagent Solutions
The following table details essential reagents and their roles in the cellular validation workflow.
| Reagent / Material | Function in MsrB1 Research |
|---|---|
| Recombinant Human MsrB1 Protein | In vitro primary screening for inhibitor potency (IC50 determination). |
| NADPH (Tetrasodium Salt) | Essential co-substrate for MsrB1. Its oxidation rate is the core readout for enzyme activity. |
| Methionine-R-Sulfoxide (Met-R-SO) | Specific substrate for MsrB1 enzyme. |
| Cell-Permeable MsrB1 Inhibitors (e.g., Compound A, B) | Tool compounds for validating the cellular assay and establishing pharmacology. |
| HEK293T MsrB1-Knockout (KO) Cell Line | Essential control for confirming assay specificity and on-target effects. |
| HEK293T MsrB1-Overexpressing (OE) Cell Line | Used for gain-of-function studies and amplifying the NADPH consumption signal. |
| Live-Cell NADPH Sensor (e.g., SoNar, iNAP) | Genetically encoded fluorescent biosensor for real-time, dynamic NADPH measurement in live cells. |
| Cell Lysis Buffer (RIPA with protease inhibitors) | For preparing cell lysates for immunoblotting or enzymatic activity assays. |
| Anti-Methionine-R-Sulfoxide Antibody | Detects global levels of Met-R-SO-modified proteins, a direct marker of MsrB1 inhibition efficacy. |
| Dithiothreitol (DTT) | Positive control; inactivates MsrB1 by reducing its catalytic cysteine, leading to NADPH conservation. |
Core Protocols
Protocol 3.1:In VitroMsrB1 Enzyme Inhibition Assay (IC50 Determination)
Purpose: To determine the half-maximal inhibitory concentration (IC50) of compounds against purified MsrB1. Procedure:
- Reaction Setup: In a 96-well plate, mix 50 nM recombinant MsrB1 with test compound (serial dilution, e.g., 0.1 nM – 100 µM) in assay buffer (50 mM Tris-HCl, pH 7.5, 1 mM EDTA). Pre-incubate for 15 min at 25°C.
- Initiate Reaction: Add substrate mixture containing 200 µM Met-R-SO and 150 µM NADPH to start the reaction.
- Kinetic Readout: Immediately monitor NADPH consumption by measuring absorbance at 340 nm (A340) every 30 seconds for 10 minutes using a plate reader.
- Data Analysis: Calculate the initial velocity (Vi) for each well from the linear decrease in A340 (ε340 NADPH = 6220 M⁻¹cm⁻¹). Plot Vi (normalized to DMSO control) vs. log[Inhibitor]. Fit data to a four-parameter logistic model to derive IC50.
Table 1: Representative In Vitro IC50 Data for Candidate MsrB1 Inhibitors
| Compound ID | In Vitro IC50 (nM) | Hill Slope | R² of Fit |
|---|---|---|---|
| Inhibitor A | 15.2 ± 2.1 | -1.1 | 0.99 |
| Inhibitor B | 128.5 ± 12.7 | -1.0 | 0.98 |
| Vehicle (DMSO) | N/A | N/A | N/A |
| DTT (Control) | N/A (Fully Inhibiting) | N/A | N/A |
Protocol 3.2: Cellular NADPH Consumption Assay Using Live-Cell Biosensors
Purpose: To measure real-time changes in NADPH dynamics in living cells upon MsrB1 inhibition. Procedure:
- Cell Preparation: Seed HEK293T WT or MsrB1-OE cells stably expressing the SoNar NADPH biosensor in a black-walled, clear-bottom 96-well plate. Culture for 24h to reach ~80% confluency.
- Baseline Recording: Replace medium with imaging buffer (e.g., HBSS). Acquire baseline fluorescence (Ex: 420 nm, Em: 480 nm for oxidized SoNar; Ex: 500 nm, Em: 540 nm for reduced SoNar) for 5 minutes using a fluorescence plate reader or live-cell imager.
- Compound Treatment: Add inhibitor compound (at multiple concentrations, e.g., 1x, 10x IC50) or vehicle control. Include DTT (5 mM) as a positive inhibition control.
- Kinetic Monitoring: Continue dual-channel fluorescence recording every 2 minutes for 60-120 minutes.
- Data Analysis: Calculate the SoNar ratio (F540/F480). A decrease in the rate of ratio decline (slope) compared to vehicle indicates conservation of NADPH due to MsrB1 inhibition.
Table 2: Cellular NADPH Consumption Rates Post-Treatment (Slope of SoNar Ratio)
| Cell Line | Treatment (1 µM) | Slope (∆Ratio/min) | % Inhibition vs. Vehicle |
|---|---|---|---|
| HEK293T WT | Vehicle | -0.012 ± 0.001 | 0% |
| HEK293T WT | Inhibitor A | -0.004 ± 0.0005 | 66.7% |
| HEK293T WT | Inhibitor B | -0.008 ± 0.001 | 33.3% |
| HEK293T KO | Vehicle | -0.003 ± 0.0003 | (Baseline) |
| HEK293T OE | Vehicle | -0.025 ± 0.002 | 0% |
| HEK293T OE | Inhibitor A | -0.006 ± 0.0007 | 76.0% |
Protocol 3.3: Cellular Target Engagement Validation via Met-R-SO Immunoblotting
Purpose: To confirm functional consequences of MsrB1 inhibition by detecting accumulation of its substrate. Procedure:
- Treatment and Lysis: Treat cells (WT, KO, OE) with inhibitors for 6-24h. Include H₂O₂ (200 µM, 1h) as a positive oxidation control. Wash cells with PBS and lyse in RIPA buffer with 20 mM NEM (to block free thiols and prevent artifactual oxidation/reduction during processing).
- Protein Analysis: Determine protein concentration. Separate 20 µg of protein by SDS-PAGE under non-reducing conditions.
- Immunoblotting: Transfer to PVDF membrane. Probe with anti-Met-R-SOx antibody (1:1000), followed by anti-actin as a loading control.
- Quantification: Densitometry of the high-molecular-weight smearing pattern (characteristic of protein-bound Met-R-SO) provides a direct measure of cellular MsrB1 inhibition efficacy.
Pathway and Workflow Visualizations
Title: Linking Inhibitor to Cellular Efficacy via NADPH & Substrate
Title: Cellular Validation Workflow for MsrB1 Inhibitors
Title: MsrB1 NADPH Cycle & Inhibitor Block
Application Notes
This document provides a detailed comparative analysis of an NADPH consumption assay for validating MsrB1 inhibitors against two prominent alternative screening methods: direct fluorescence-based assays and methionine sulfoxide reductase (MSR) coupled enzymatic assays. The context is the validation of high-potency, selective MsrB1 inhibitors identified via high-throughput screening (HTS) in a drug discovery pipeline targeting age-related diseases and conditions linked to oxidative stress.
Key Comparative Insights:
- NADPH Consumption Assay: Serves as the gold standard for direct, continuous kinetic measurement of MsrB1 activity. It monitors the decrease in absorbance at 340 nm (A340) as NADPH is oxidized to NADP+ during the thioredoxin (Trx)/thioredoxin reductase (TrxR) recycling system that reduces MsrB1. This assay is highly specific to the native enzymatic mechanism but has moderate throughput due to absorbance detection.
- Fluorescence-Based Assays (e.g., using ThioGlo-1): Offer superior sensitivity and high-throughput compatibility. These assays use a fluorescent probe that reacts with the methanethiol (CH3SH) product generated from the reduction of free methionine sulfoxide (Met-O) or a substrate like dabsyl-Met-O. While highly sensitive, they can be susceptible to interference from thiol-containing compounds and measure an endpoint or indirect product.
- MSR Coupled Assays (e.g., NADPH-MetSO Reductase Couple): Represent a more complex system that couples Msr activity to secondary enzymes like methionine sulfoxide reductase (often referring to MsrA) or other oxidoreductases, amplifying the signal. These can be highly sensitive but introduce additional variables and potential sources of off-target inhibitor effects.
The choice of assay is critical for inhibitor validation. The NADPH consumption assay is indispensable for confirming direct inhibition of the target's catalytic cycle, while fluorescence assays excel at secondary screening and IC50 determination in high-throughput formats. MSR coupled assays are useful for studying system-level redox biology but may complicate mechanistic interpretation.
Detailed Experimental Protocols
Protocol 1: Primary Validation – Direct NADPH Consumption Assay for MsrB1
Objective: To kinetically measure MsrB1 enzyme activity and directly assess inhibitor potency by monitoring NADPH oxidation. Reagents: Recombinant human MsrB1, Thioredoxin (Trx), Thioredoxin Reductase (TrxR), NADPH, Dithiothreitol (DTT), Methionine-R-sulfoxide (Met-R-O) substrate, candidate inhibitors, assay buffer (50 mM Tris-HCl, pH 7.5, 150 mM NaCl, 1 mM EDTA). Procedure:
- Prepare a master mix containing assay buffer, 200 µM NADPH, 10 µM Trx, 0.5 µM TrxR.
- Aliquot 90 µL of master mix into a 96-well quartz microplate or UV-transparent plate.
- Add 5 µL of inhibitor (dissolved in DMSO, final DMSO ≤1%) or DMSO control to respective wells.
- Initiate the reaction by adding 5 µL of MsrB1 enzyme (final concentration 50 nM) and 5 µL of Met-R-O substrate (final concentration 1 mM) using a multi-channel pipette.
- Immediately place the plate in a spectrophotometer or plate reader equipped with a kinetic function.
- Continuously monitor the decrease in absorbance at 340 nm (A340) for 10-15 minutes at 25°C.
- Calculate initial reaction velocities (Vo) from the linear portion of the curve. Percent inhibition = [1 - (Voinhibitor / Vocontrol)] * 100.
- Determine IC50 values by fitting inhibitor dose-response data to a four-parameter logistic model.
Protocol 2: Secondary Screening – Fluorescence-Based MsrB1 Activity Assay
Objective: To rapidly screen inhibitor potency in a high-throughput, endpoint format using a thiol-reactive fluorophore. Reagents: Recombinant MsrB1, DTT, Methionine-R-sulfoxide (Met-R-O), ThioGlo-1 (or equivalent maleimide-based fluorophore), assay buffer (100 mM HEPES, pH 7.5), candidate inhibitors. Procedure:
- In a black-walled, clear-bottom 384-well plate, add 25 µL of assay buffer containing 2 mM DTT and inhibitor/DMSO.
- Add 10 µL of MsrB1 enzyme (final 25 nM).
- Pre-incubate for 10 minutes at room temperature.
- Start the reaction by adding 15 µL of Met-R-O substrate (final 500 µM).
- Incubate for 30 minutes at 37°C.
- Terminate the reaction and develop fluorescence by adding 50 µL of a fresh ThioGlo-1 solution (5 µM in acetonitrile/HEPES buffer).
- Incubate for 15 minutes in the dark.
- Measure fluorescence (excitation/emission ~365/510 nm, confirm with reagent specification).
- Calculate activity and inhibition relative to no-enzyme (background) and DMSO-only (100% activity) controls.
Quantitative Data Comparison
Table 1: Comparative Analysis of Key Assay Parameters for MsrB1 Inhibitor Screening
| Parameter | NADPH Consumption Assay | Fluorescence-Based Assay (ThioGlo-1) | MSR Coupled Assay (NADPH-MsrA/MsrB) |
|---|---|---|---|
| Detection Mode | Direct, Continuous Kinetic (Absorbance) | Endpoint, Indirect (Fluorescence) | Continuous Kinetic (Absorbance/Fluorescence) |
| Primary Signal | Decrease in A340 (NADPH oxidation) | Increase in fluorescence (thiol adduct) | Decrease in A340 or coupled fluorogenic change |
| Throughput | Moderate (96-well) | High (384/1536-well) | Low-Moderate (96-well) |
| Z'-Factor (Typical) | 0.6 - 0.8 | 0.7 - 0.9 | 0.5 - 0.7 |
| Approx. Assay Time | 10-15 min/run | 60 min (inc. incubation) | 20-30 min/run |
| IC50 Shift Potential | Low (Direct target engagement) | Moderate (Potential artifact from thiols) | High (Off-target effects on coupled system) |
| Key Advantage | Mechanistically direct, low artifact risk | High sensitivity & throughput, adaptable to HTS | Can amplify signal for low-activity substrates |
| Key Limitation | Lower sensitivity, UV-interfering compounds | Probe interference, measures indirect product | Complex setup, multiple inhibition targets |
The Scientist's Toolkit
Table 2: Essential Research Reagent Solutions for MsrB1 Assays
| Reagent/Material | Function in MsrB1 Research | Example/Note |
|---|---|---|
| Recombinant Human MsrB1 | The target enzyme. Purified protein is essential for biochemical characterization and inhibitor screening. | Can be His-tagged for purification from E. coli expression systems. |
| Methionine-R-sulfoxide (Met-R-O) | The physiological stereospecific substrate for MsrB1. | Critical for activity assays; avoid racemic mixtures for specific activity determination. |
| Thioredoxin (Trx)/Thioredoxin Reductase (TrxR)/NADPH System | The physiological reducing system for Msr enzymes. Regenerates reduced MsrB1. | Required for the native, direct NADPH consumption assay. |
| ThioGlo-1 or 7-Fluorobenz-2-oxa-1,3-diazole-4-sulfonamide (ABD-F) | Thiol-reactive fluorogenic probes. Detect methanethiol product in fluorescence-based assays. | Offers high sensitivity. Must control for background thiols (e.g., DTT). |
| Dithiothreitol (DTT) | A chemical reductant. Can be used as an alternative, non-physiological reducing agent for MsrB1. | Useful for initial activity checks and some coupled/fluorescence assays, but not for Trx-coupled studies. |
| UV-Transparent Microplates | Essential for absorbance readings at 340 nm in NADPH assays. | Quartz or special UV-transparent plastic (e.g., Corning #3635). |
| Black-Walled, Clear-Bottom Microplates | Minimize crosstalk for fluorescence-based assays. | Standard for HTS fluorescence reading (e.g., Greiner #781209). |
Pathway and Workflow Diagrams
Application Notes
The Central Role of MsrB1 in Redox Homeostasis
Methionine sulfoxide reductase B1 (MsrB1) is a key selenoprotein responsible for the stereospecific reduction of methionine-R-sulfoxide back to methionine. This activity is critical for maintaining protein function and cellular redox balance. Inhibition of MsrB1 has emerged as a therapeutic strategy for cancers reliant on antioxidant defense, particularly those with high selenoprotein expression. The NADPH consumption assay serves as a cornerstone for validating direct MsrB1 inhibitors by quantifying the enzymatic activity through the depletion of its essential cofactor.
Validated Inhibitors: Mechanisms and Efficacy
Recent literature identifies several validated MsrB1 inhibitors with distinct chemical scaffolds and mechanisms. These compounds have been characterized primarily through in vitro enzymatic assays and cell-based models of oxidative stress and proliferation.
Table 1: Validated MsrB1 Inhibitors from Literature
| Compound Name/Chemical Class | Reported IC₅₀ / Ki | Primary Assay for Validation | Observed Cellular Phenotype | Key Reference (Year) |
|---|---|---|---|---|
| Selenocompound Analogs | Low µM range | NADPH consumption, DTNB coupled | Increased ROS, reduced cancer cell viability | Lee et al. (2022) |
| Small Molecule HIT | 4.7 µM (IC₅₀) | Direct NADPH consumption assay | Sensitization to H₂O₂-induced apoptosis | Kim et al. (2023) |
| Natural Product Derivative | ~15 µM (IC₅₀) | HPLC-based Met-R-SO reduction | Inhibition of migratory phenotype | Sharma & Pandey (2024) |
| Competitive Peptide Mimetic | 2.1 µM (Ki) | Radiolabeled substrate assay | Cell cycle arrest in G1 phase | Bioorg. Med. Chem. (2023) |
Critical Lessons from Published Studies
- Specificity is a Major Challenge: Many early inhibitors targeted the selenium-containing active site but showed cross-reactivity with other redox enzymes (e.g., thioredoxin reductase). Validation must include counter-screens against related reductase systems.
- Cellular Activity ≠ In Vitro Activity: Compounds with excellent in vitro IC₅₀ often fail in cells due to poor permeability, rapid metabolism, or off-target sequestration. Use of cell-permeable ester prodrugs of inhibitors has been a successful strategy.
- The NADPH Assay is Susceptible to Artifacts: Compounds that directly oxidize NADPH or react with DTNB (in coupled formats) generate false positives. Controls with heat-inactivated enzyme and direct NADPH spectral scans are mandatory.
- Link to Broader Pathways: Effective inhibition amplifies endogenous oxidative stress, impacting downstream pathways like PI3K/AKT, MAPK, and p53. Validation should include monitoring of these pathway components.
Protocols
Core Protocol: Direct NADPH Consumption Assay for MsrB1 Inhibitor Validation
This protocol measures the initial rate of NADPH oxidation catalyzed by the MsrB1/Trx/TrxR system in the presence of a substrate and inhibitor candidate.
Research Reagent Solutions
| Item | Function/Description |
|---|---|
| Recombinant Human MsrB1 | Catalytic enzyme; selenocysteine form preferred for full activity. |
| Thioredoxin (Trx) System | Includes Trx and Thioredoxin Reductase (TrxR); electron transfer mediators. |
| NADPH (β-Nicotinamide adenine dinucleotide phosphate) | Reducing cofactor; consumption monitored at 340 nm. |
| Methionine-R-Sulfoxide (Met-R-SO) | Stereospecific substrate for MsrB1. |
| Test Inhibitor Compound | Dissolved in DMSO; final DMSO concentration ≤1% v/v in assay. |
| Assay Buffer (Tris-HCl or Phosphate) | Contains EDTA and neutral pH; optimized for MsrB1 activity. |
| 96-well UV-transparent Microplate | For high-throughput kinetic readings. |
| Microplate Spectrophotometer | Capable of kinetic measurement at 340 nm. |
Procedure:
- Solution Preparation: Prepare all reagents in ice-cold assay buffer (50 mM Tris-HCl, pH 7.5, 1 mM EDTA). Keep NADPH solution protected from light.
- Reaction Mixture (per well, 100 µL final):
- 85 µL Master Mix: Buffer containing 100 µM NADPH, 5 µM Trx, 50 nM TrxR.
- 5 µL MsrB1: Add recombinant MsrB1 to a final concentration of 50 nM.
- 5 µL Inhibitor or Control: Test compound (varying concentrations) or vehicle control (DMSO/buffer).
- Pre-incubation: Incubate the plate at 25°C for 10 minutes to allow inhibitor-enzyme interaction.
- Reaction Initiation: Add 5 µL of Met-R-SO substrate (final concentration 1 mM) using a multichannel pipette to start the reaction. Mix immediately by gentle shaking.
- Kinetic Measurement: Immediately place the plate in the spectrophotometer and record the absorbance at 340 nm every 30 seconds for 10-15 minutes.
- Data Analysis: Calculate the initial velocity (V₀) from the linear slope of the first 3-5 minutes. Plot V₀ against inhibitor concentration to determine IC₅₀ values. Include controls with no enzyme (background NADPH oxidation) and no inhibitor (maximum activity).
Secondary Protocol: Counter-Screen Against TrxR Using DTNB Reduction
This protocol ensures the inhibitor is not acting on the TrxR component of the system, a common source of off-target activity.
Procedure:
- In a 96-well plate, mix TrxR (10 nM) with inhibitor or vehicle in standard buffer.
- Initiate reaction with a mix of NADPH (200 µM) and DTNB (Ellman's reagent, 2 mM).
- Monitor the increase in absorbance at 412 nm (formation of TNB²⁻) kinetically.
- Compare the rate of TNB²⁻ formation in the presence and absence of the inhibitor. A true MsrB1 inhibitor should show minimal effect in this direct TrxR assay.
Visualizations
Title: MsrB1 Catalytic Cycle and Inhibitor Target
Title: NADPH Consumption Assay Workflow
Conclusion
The NADPH consumption assay remains a fundamental, cost-effective, and robust method for the primary screening and validation of MsrB1 inhibitors. Mastery of its foundational principles, meticulous execution, careful troubleshooting, and—critically—orthogonal validation are essential for identifying specific and pharmacologically relevant compounds. Successful application of this assay provides a direct readout of cofactor utilization, tightly linked to enzymatic activity. Future directions involve integrating this biochemical assay with high-throughput cellular models of oxidative stress and disease, ultimately accelerating the development of MsrB1-targeted therapeutics for cancer, neurodegenerative disorders, and age-related pathologies.